 |
Understanding ONH Dynamics in Glaucoma and Beyond
Clinicians must know what a healthy optic nerve head looks like and how it functions before they can appreciate the dysfunction that happens with disease.
By Catherine Hogan, OD, and Andrew Rixon, OD
Release Date: July 15, 2021
Expiration Date: July 15, 2024
Estimated Time to Complete Activity: 2 hours
Jointly provided by Postgraduate Institute for Medicine (PIM) and Review Education Group
Educational Objectives: After completing this activity, the participant should be better able to:
- Discuss the pathophysiology of optic nerve head disorders.
- Explain how the optic nerve head functions.
- Identify potentially suspicious optic nerve pathology.
- Diagnose various optic nerve head disorders.
Target Audience: This activity is intended for optometrists engaged in eye care of the optic nerve head.
Accreditation Statement: In support of improving patient care, this activity has been planned and implemented by the Postgraduate Institute for Medicine and Review Education Group. Postgraduate Institute for Medicine is jointly accredited by the Accreditation Council for Continuing Medical Education, the Accreditation Council for Pharmacy Education, and the American Nurses Credentialing Center, to provide continuing education for the healthcare team. Postgraduate Institute for Medicine is accredited by COPE to provide continuing education to optometrists.
Reviewed by: Salus University, Elkins Park, PA
Faculty/Editorial Board: Catherine Hogan, OD, and Andrew Rixon, OD
Credit Statement: This course is COPE approved for 2 hours of CE credit. Activity #122080 and course ID 73298-GL. Check with your local state licensing board to see if this counts toward your CE requirement for relicensure.
Disclosure Statements:
Authors: Drs. Hogan and Rixon have no financial interests to disclose.
Managers and Editorial Staff: The PIM planners and managers have nothing to disclose. The Review Education Group planners, managers and editorial staff have nothing to disclose.
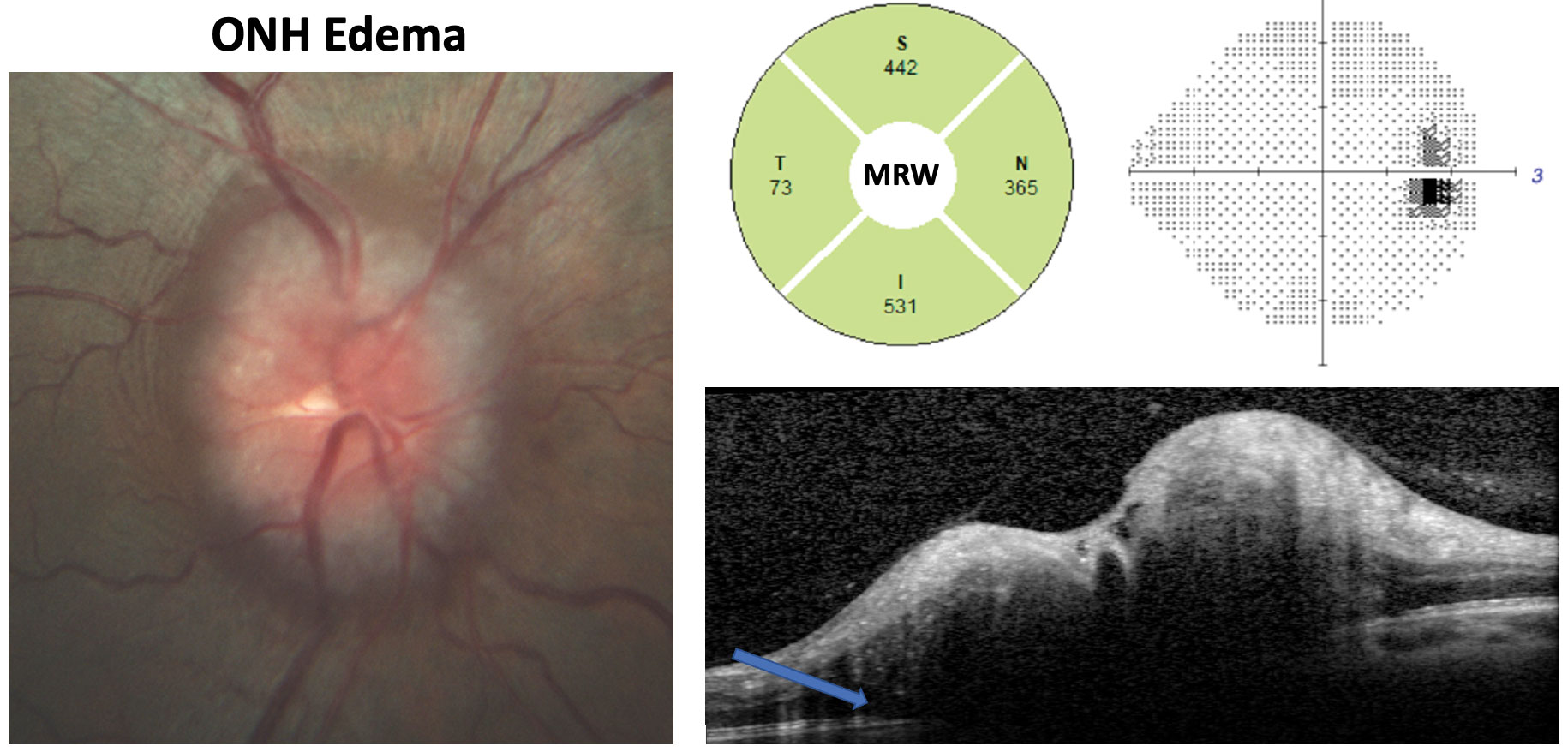 |
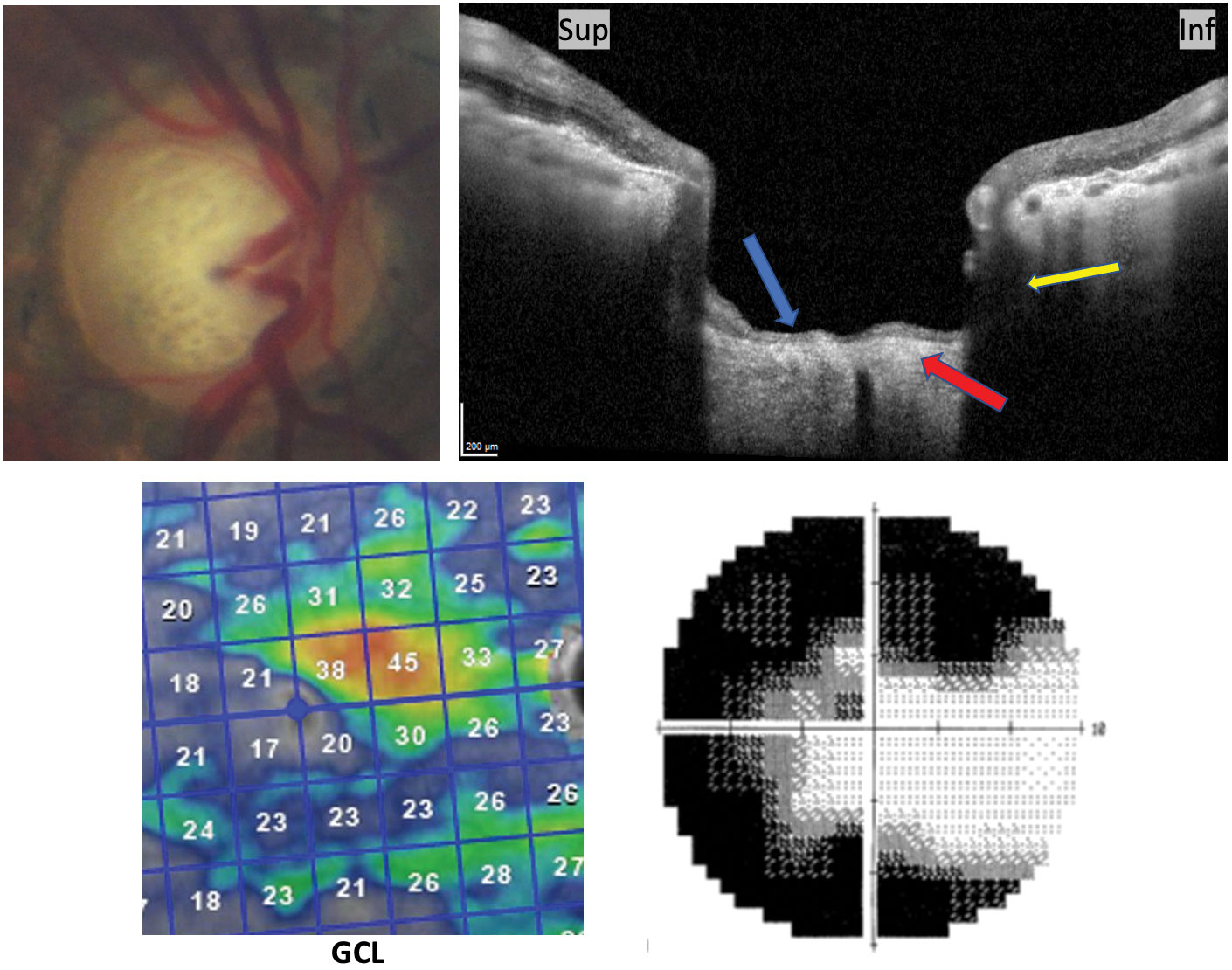
| This IIH patient has an opening ICP of 42cm H2O. Fundus photography shows Frisen grade 3 edema. Note the substantially increased MRW, enlarged blind spot on the grayscale plot and the OCT cross-section showing subretinal hyporeflective space (blue arrow) consistent with ONH edema. Click image to enlarge. |
Glaucoma is the most common optic neuropathy found in adulthood, and the most prevalent form—primary open-angle glaucoma (POAG)—is projected to affect 7.32 million individuals in the United States by 2050.1 This estimation is staggering and should encourage ODs to not become complacent or make assumptions about glaucoma diagnosis, especially as the condition is a diagnosis of exclusion.
A litany of non-glaucomatous entities can also cause optic nerve dysfunction, and although this fact confounds our job as clinicians, understanding fundamental facets of the optic nerve and employing a systematic diagnostic approach will help guide us to the truth, ultimately maximizing the patient experience.
This review will discuss the pathophysiology and clinical assessment of common optic nerve head (ONH) disorders with a practical emphasis on their differentiating features.
A Systematic Approach
Clinically, a detailed and systematic stereoscopic analysis of the ONH and its surrounding tissue using the slit lamp and various condensing lenses provides framework for the assessment. Although quantitative variables are important, attention to the qualitative aspects and morphology of the ONH should be the main point of emphasis, as there is considerable quantitative overlap between normal and unhealthy ONHs.5
Understanding how to evaluate the glaucomatous disc is key to differentiating the disease. One of the most repeatable systems you can follow for guidance is the ‘five Rs’ developed as part of FORGE (Focusing Ophthalmology on Reframing Glaucoma Evaluation): (1) observing the scleral ring to identify the boundaries of the optic disc and determine its size (larger vertical disc diameters will naturally have larger cups), (2) assessing the retinal nerve fiber layer (RNFL) for localized or diffuse loss, (3) examining the neuroretinal rim while specifically evaluating the width, shape and color in all sectors with the premise that on average the disc will follow the ISNT rule and will not have focal tissue loss, (4) checking for retinal or disc hemorrhages, which often straddle the rim but can be found at the level of the lamina or in the peripapillary retina and (5) evaluating for a region of peripapillary atrophy (PPA) of chorioretinal tissue particularly adjacent to areas where the neuroretinal rim is thinner.5,6 An additional glaucoma feature to be on the lookout for is deep or laminar cupping created by tissue remodeling and subsequent excavation beneath the scleral canal.7-9
A Closer Look at the ONHRecognizing what an ONH looks like and how it functions allows us to better discriminate between normal and abnormal findings, thus unlocking the ability to more specifically diagnose diseases of the ONH. Functionally, the optic nerve transmits stimuli between the neurosensory retina and the lateral geniculate body and cerebral visual pathways. The ONH refers to the ophthalmoscopically visible anterior surface of the nerve. The ONH consists primarily of four components: neural fibers, glial cells, extracellular matrix support and vascular tissue.2-4 The neural tissue is formed by the retinal ganglion cell axons of the RNFL, originating primarily in the macula and converging radially on the ONH in arcuate bundles, forming a convex periphery and the first layer of the ONH, the surface nerve fiber layer. This neural tissue appears orange-pink on fundoscopy as a result of the reflectance of light off of the dense capillary network that supplies the disk surface. These fibers then bend abruptly, passing through the second layer—the prelaminar layer—which is made up of loose, capillary-containing glial tissue. The previously mentioned fiber bend creates the central and concave cup, which appears yellow as a result of the supporting collagenous fibers found in the third layer, the lamina cribrosa region. The collagenous lamina cribrosa is made up of fenestrated connective tissue lamellae that contain capillaries and are covered in astrocytes. The nerve fiber layer passes through the scleral lamina cribrosa and the fourth layer, the retrolaminar region, prior to exiting the globe.2,3 The blood supply to the optic nerve is mainly from the posterior ciliary artery, with the exception of the surface of the nerve which receives its supply from retinal circulation. Importantly, ONH blood supply is sectoral in nature.4 Knowing how the neural and vascular tissue is distributed is critical in recognizing how characteristic patterns of tissue damage can occur in various ONH disorders. |
Employing a systematic method can help minimize diagnostic errors. Studies have shown that general ophthalmologists are twice as likely to underestimate glaucoma compared with glaucoma subspecialists. ONHs most likely to be underestimated were horizontally ovoid in shape and those with moderate PPA. Factors associated with underestimation were failure to correctly grade vertical cup-to-disc (C/D) ratios, assess disc hemorrhages and capture RNFL and rim loss.
ONHs most likely to be identified as glaucomatous when they were not were large and had nerves with horizontal or vertical disc tilt, among other factors.10 Researchers found that in a group of optometric observers, average-sized nerves were correctly identified 90% of the time. However, large ONHs tended to be misidentified as glaucomatous, and even more problematically, small ONHs associated with glaucoma were misidentified as normal, reinforcing the size bias during ONH evaluation that we all need to remain aware of.11
These findings may also be influenced by the lack of structured funduscopic evaluation. A study monitoring the gaze behavior of trainees and experts when assessing ONHs found that trainees, although they spent more time evaluating ONHs, exhibited no specific gaze pattern regardless of whether the ONHs had suspicious findings or not and mainly concentrated on the optic cup. Experts, however, took less time and were more systematic and consistent in their approach, concentrating on areas of interest (rim tissue, RNFL and superior and inferior poles) and suspicious structural findings as they presented.12
How does optic nerve damage occur in glaucoma? At present, we know that a number of factors influence the retinal ganglion cells and ONH. These include intraocular pressure (IOP), cerebrospinal fluid pressure (CSFP) and ocular perfusion pressure (OPP).13 The complex interrelations among these factors result in glaucomatous optic neuropathy. Although there is not currently a universal consensus of the exact relationship between these factors, there is a characteristic pattern of glaucomatous ONH damage that is known, helping to more clearly delineate glaucoma from other ONH disorders.
In essence, glaucoma is a laminopathy, whereas other optic neuropathies are not. The aforementioned term “cupping” can create confusion as it can be conflated with C/D ratio. In fact, cupping has been proposed to involve two components: prelaminar and laminar.8 Prelaminar cupping affects prelaminar neural tissue, and progressive loss of that tissue will increase the depth and the width of the cup, notably increasing the C/D ratio.
The following is where the distinction between non-glaucoma and glaucomatous cupping occurs. Multiple in vivo and cadaveric studies have shown that what separates glaucomatous ONH damage from other neuropathies is the involvement of both connective tissue and neural tissue. Glaucomatous optic nerves undergo progressive posterior bowing/displacement, disorganization and deformation of the lamina cribrosa and peripapillary scleral connective tissue, in addition to the loss of retinal ganglion cell axons.7-9,14 This results in the laminar or deep cupping consistent with glaucoma.
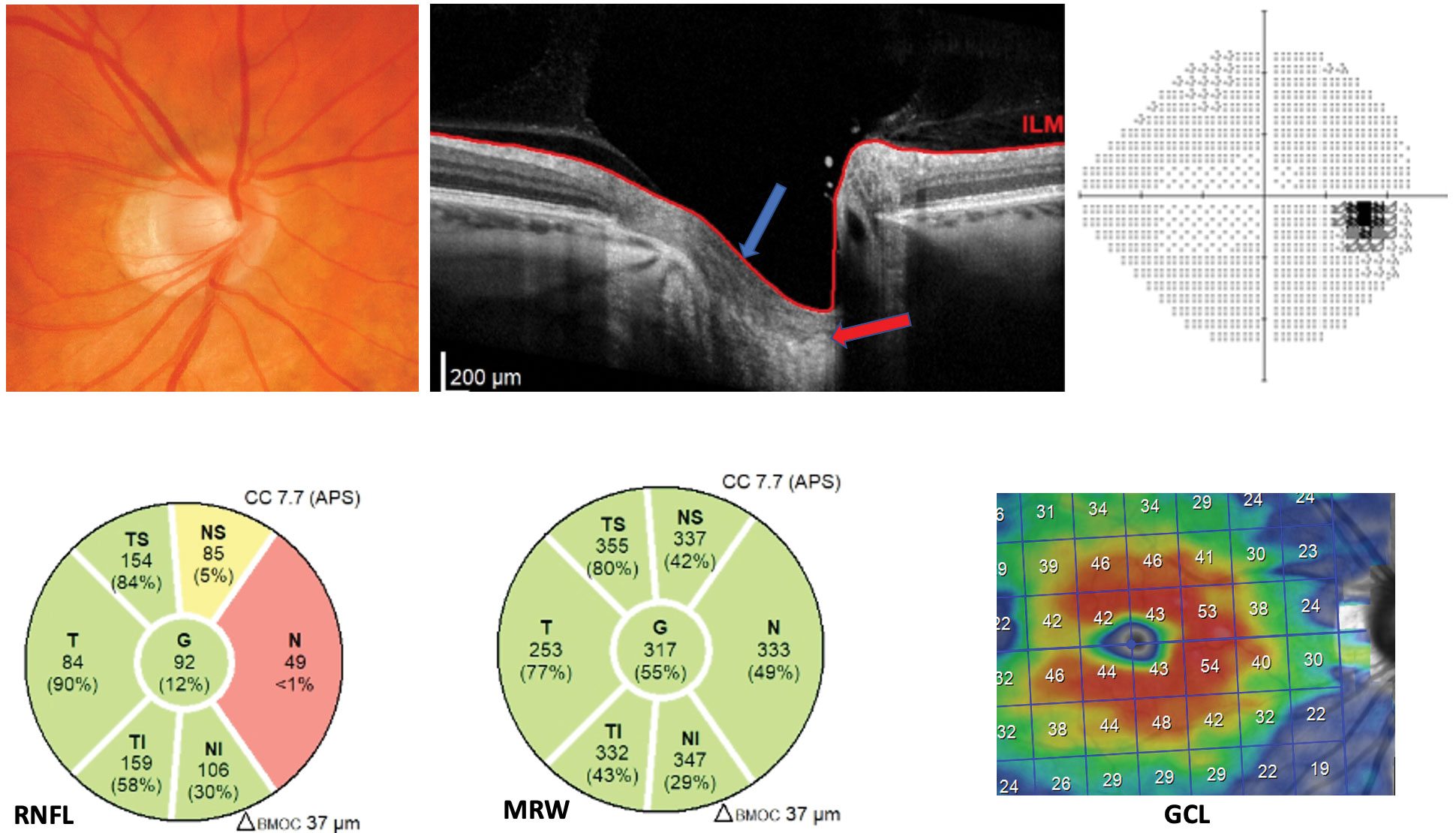 |
| This -7.00D myope was previously classified as a glaucoma suspect. Note the malinsertion and intact pre-laminar tissue (blue arrow) found on the ONH cross-section. The lamina is marked by the red arrow. Additionally, the MRW is unremarkable and the nasal RNFL tissue is thin secondary to temporalized vasculature. Both the ganglion cell layer and the visual field are unremarkable. Click image to enlarge. |
Clinical Differentiation
Recognizing the morphologic changes found in glaucoma provides insight for differentiating the disease from other ONH disorders. These differential diagnoses can range from anatomical optic nerve disorders that clinically confound the observer to various neuropathies that cause optic nerve damage via other pathophysiological pathways. Using a combination of detailed clinical examination and ancillary testing will allow the optometric physician to make an informed and accurate diagnosis, treatment and management plan for the patient.9 This all begins with differentiating glaucoma from its masqueraders, some of which we cover here:
Large optic disc. At first glance, a large optic disc with a correlating large cup commonly leads to glaucoma misdiagnosis. Looking for glaucomatous features instead of zeroing in on cup size alone is an important shift in the process.15 First, consider the patient’s demographics. Males and African Americans are more likely to have anatomically larger discs.16 Measure the vertical height of the optic disc during funduscopy, and apply the correction factor for magnification per the dioptric power of the lens used. For instance, a Volk Super 66D lens or digital 1.0x offers a 1.0 correction factor compared with the Volk 90D lens’ correction factor of 1.3.17,18 Large disc height will suggest large disc area, a presentation which can be misdiagnosed as glaucoma.5 Disc area values are populated and confirmed by most OCT modalities.19
Compare quadrants of rim tissue to better recognize how in a large disc, even with a large cup, the rim tissue can be intact. Assess the optic nerve and arcades using the red-free filter to better highlight the RNFL and confirm the lack of wedge defects, a progressive neuropathic disease finding wherein focal RNFL damage extends from the optic disc and into the papillomacular bundle.20
Look within the cup to assess the lamina cribrosa; normal cups not under IOP stressors may have visible laminar dots but should not have laminar deformation/reconfiguration. The clinical cupping associated with glaucoma involves permanent and progressive prelaminar thinning and laminar deformation as a result of IOP stress. This pathophysiologic damage over time is absent in anatomically large healthy discs.9
Gauging stability and confirming there is no progressive loss of rim tissue when dealing with anatomically large discs can be accomplished with multiple technologies. RNFL thickness, ganglion cell analysis (GCA) and visual field results will remain stable over time in patients with large healthy discs. However, RNFL thickness will be increased in superior, inferior and global quadrants in large discs.21 Spectral-domain (SD) RNFL analysis will also confirm disc area and compare the measured area (in mm2) to a database of normative values. One study used confocal scanning laser ophthalmoscopy (CSLO) and SD-OCT to define a large disc area as greater than 2.43mm2.15
Analysis of the rim area also provides contrast between anatomically large discs and glaucoma. Using CSLO, it has been found that progressive glaucomatous optic nerves experience global rim area loss 3.7 times faster than large healthy optic nerves.22 OCT analysis of Bruch’s membrane opening-minimum rim width (BMO-MRW) measures the minimum distance from the BMO to the internal limiting membrane, allowing for more accurate assessment of the neuroretinal rim regardless of disc size.15,23 It is reported that larger disc areas physiologically have larger BMO areas, which contributes to thinner but stable BMO-MRW results. Therefore, guided progression analysis of RNFL and BMO-MRW is paramount to classify stability vs. progression over time.15,24
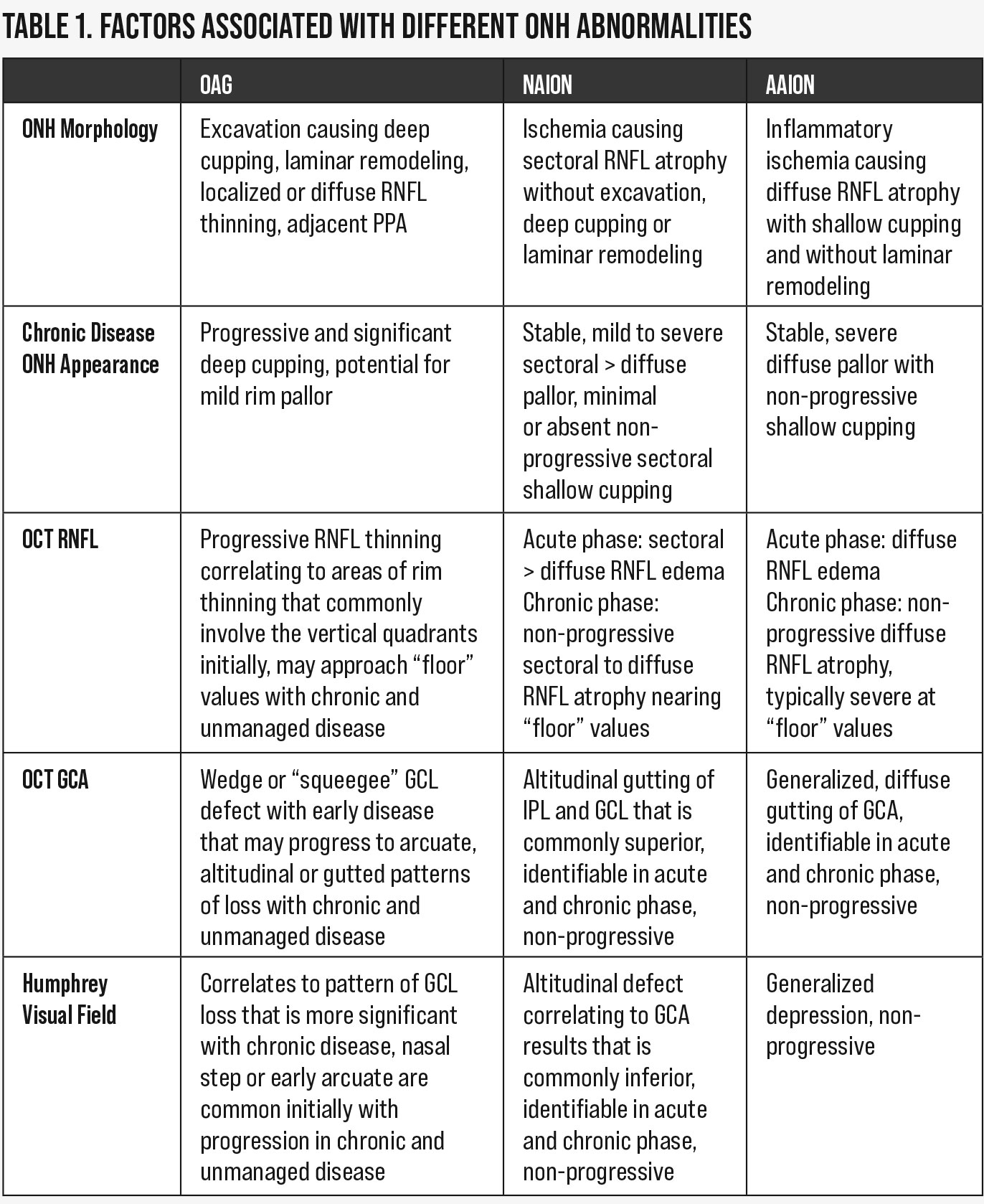 |
| Table 1. Factors associated with different ONH abnormalities. Click table to enlarge. |
Tilted disc syndrome (TDS). This congenital condition is another anomalous optic nerve condition. It arises from malclosure of the embryonic optic fissure during the second month of gestation. This presents clinically with inferonasally rotated optic discs in tandem with juxtapapillary changes and retinal vascular abnormalities, including adjacent inferonasal crescent or PPA, retinal pigment epithelium and choroidal thinning, posterior staphyloma and situs inversus. The optic disc rotation is non-progressive but can impair the clinician’s ability to visualize the true boundaries of the rim tissue and C/D ratio. The anatomical insertion may lead to a sense of sectoral cupping, but physiologically TDS does not exemplify rim thinning or laminar deformation.9,25,26
OCT sheds light on what can be clinically confounding when differentiating TDS from glaucoma. RNFL results associated with TDS depend on optic nerve morphology and torsion degree but typically show false RNFL thinning in nasal quadrants, which are stable over time on glaucoma progression analysis (GPA).
GCA and BMO-MRW provide better specificity than RNFL for TDS evaluation. GCA has more uniformity, with less confounding results due to anatomy, and shows intact, stable ganglion cell structure over time. BMO-MRW adjusts for individual ONH rim tissue orientation and in TDS, reveals intact, stable neuroretinal rim tissue over time.27
Visual field defects in TDS are most commonly found in the superior temporal quadrant (recall that inferonasal is the most common disc rotation), with reports of up to 33%.28,29 These defects are non-progressive and do not necessarily respect the horizontal meridian, emphasizing how TDS physiology spares the nerve fiber layer and lamina cribrosa of thinning.26,28,29
Pseudopapilledema and ONH drusen (ONHD). The term pseudopapilledema encompasses clinical variations of anomalous ONH elevation, blurring or an indistinct margin appearance in the confirmed absence of RNFL edema. Congenital anomalies including tilted and malinserted discs, hypoplasia, myelinated nerve fiber layers, Bergmeister’s papillae and ONHD may create the clinical appearance of an indistinct optic nerve, leading the primary and emergent differential diagnosis to be true optic disc edema.30
Pseudopapilledema impairs complete visualization of rim tissue. Serial fundus photography, visual field testing, GCA and OCT with BMO-MRW are valuable tools that, when assessed altogether, provide better visualization of the anomalous optic nerve structure and a baseline determination of function.27 If GPA signals progress over time, the clinician will be able to identify glaucomatous patterns of change that may have been otherwise masked by the anomalous clinical appearance.30,31
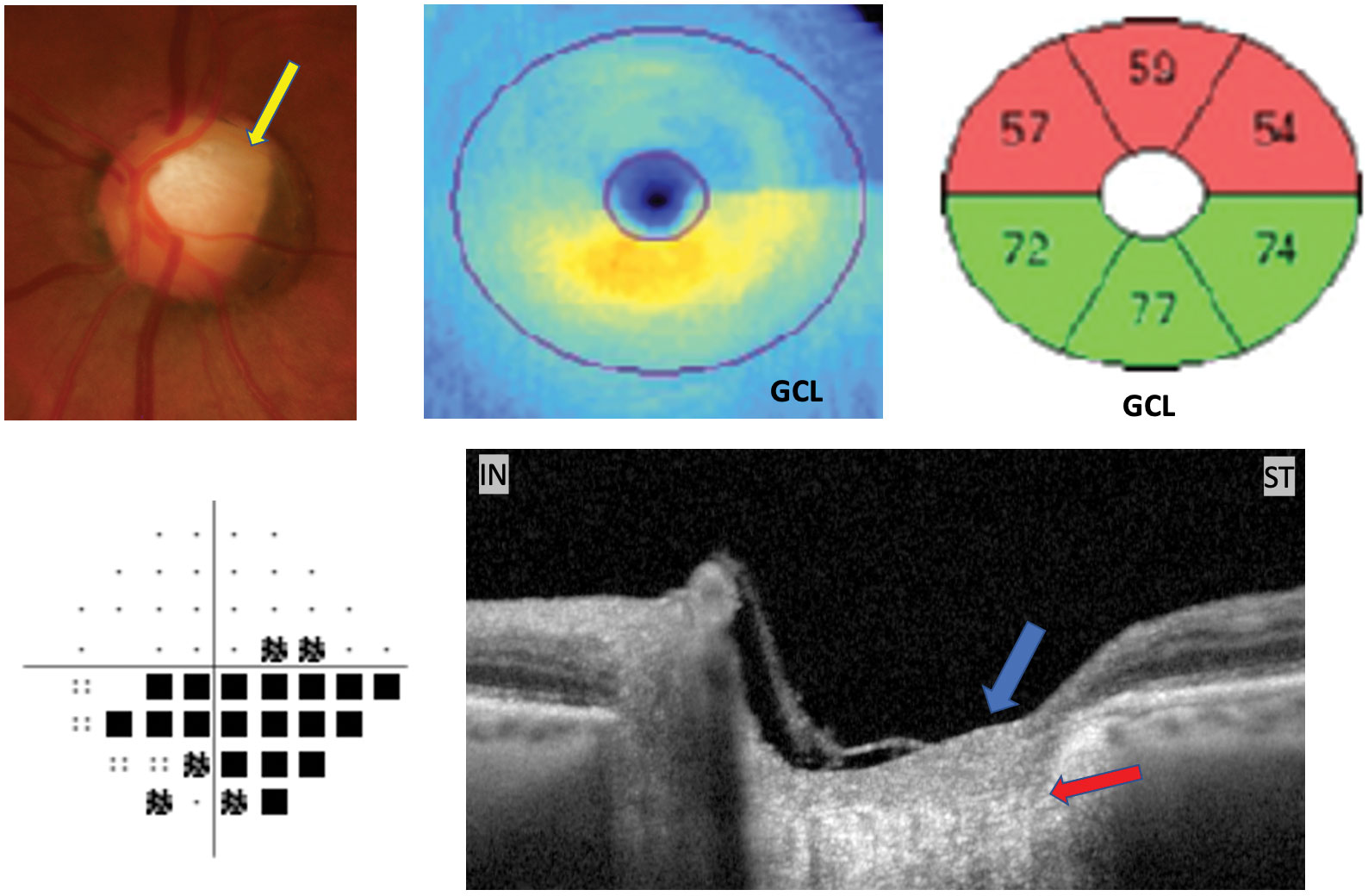 |
| This patient was previously treated for moderate primary OAG OU. They exhibit superior temporal sectoral pallor (yellow arrow), hemispheric ganglion cell loss, inferior visual field defect and intact prelaminar (blue arrow) and laminar tissue (red arrow) shown on OCT through the superior temporal rim. This is consistent with AION, not glaucoma. Click image to enlarge. |
ONHD is considered a subtype of pseudopapilledema and presents with acellular deposits consisting of calcium, amino and nucleic acids and mucopolysaccharides that form in the prelaminar ONH.32 Caucasians and patients with a family history of ONHD have a higher prevalence, and the majority of ONHD cases are bilateral.30,33 Researchers reported the best supported understanding of ONHD formation by using histological studies to determine the presence of abnormal axonal metabolism, leading to axonal disruption, extrusion of mitochondria into the prelaminar extracellular space and dysregulation of calcium deposition.32,33
Another study determined the angioarchitecture in ONHD differs from the normal optic nerve. The authors report that embryologic development of optic nerve vasculature is disturbed early on by the presence of ONHD, creating risk of vascular flow impedance at the level of the central retinal artery and its arterial bifurcations within the optic disc. Thus, while ONHD is considered a benign condition, there may actually be risk of vascular obstruction at the optic nerve, inducing chronic hypoxia and long-term RNFL damage.33,34
ONHD and glaucoma may present concurrently. Therefore, the first step in clinical evaluation is a thorough examination of the optic nerve, its vasculature and the surrounding RNFL. The more superficial the ONHD, the more “lumpy-bumpy” the disc margin, with a nodular, yellow-to-gray papillary color. Be aware that the peripapillary retinal vessels are never obscured in benign ONHD—an important distinction from papilledema. The indistinct or elevated disc margins caused by ONHD can mask the appearance of glaucomatous cupping, making funduscopic examination challenging. As a result, assessment for wedge defects with the red-free filter may be the best funduscopic clue for concurrent glaucoma.30,33
Confirmation of ONHD is a necessity, as misdiagnosis may burden the patient with costly over-management and needless treatment. Recognizing the presence of buried and/or superficial ONHD with appropriate technological work-up will not only rule out emergent diagnoses like papilledema but will also assist in the identification of non-glaucomatous vs. glaucomatous patterns. Fundus autofluorescence (FAF) of the optic nerve will show hyper-autofluorescent, superficial, calcified drusenoid bodies. FAF sensitivity to detect buried ONHD is reported to be as low as 12% to 18%.33
B-scan ultrasonography, the traditional gold standard for ONHD identification, will reveal hyper-echoic drusenoid bodies within the optic nerve, but it also has a lower detection rate for buried drusen, likely because they are less calcified.35 So, the clinician may need enhanced-depth imaging OCT (EDI-OCT) or swept-source OCT (SS-OCT) of the optic nerve to visualize buried drusen.33,36 EDI-OCT and SS-OCT have been suggested as the new gold standard for visualizing superficial and buried ONHD, which appear as hyporeflective bodies with surrounding hyper-reflective borders.36
With confirmation of ONHD, the next step is to rule out neuropathic or glaucomatous damage. RNFL thinning is typically localized to the area with greatest ONHD aggregation. The nasal quadrant is the most likely affected section, and RNFL thinning stabilizes as the superficial movement of ONHD concludes. Progressive RNFL thinning over time, especially in the inferior and superior quadrants, may signal glaucomatous rim thinning.
Monitoring the ganglion cell layer (GCL) is the most sensitive measure to examine for glaucomatous neuro-retinal damage, as benign ONHD will not cause ganglion cell deficits.33 Visual field results may be normal (48%), but defects can occur, particularly in superficial ONHD, with varying patterns including enlarged blind spot (30%) and incomplete arcuate or concentric narrowing defect (22%).33,37 The slow, progressive nature of visual field defects in ONHD is debated, with etiologies of mechanical compression or vascular compromise currently under investigation.33,36
While ONHD pathophysiology differs from that of glaucoma, the ONHD structure-function relationship may involve ONH hypoperfusion and symptomatic visual deficiencies. Currently, there is no standard treatment for ONHD. Observation over time is preferred with the understanding that elevated IOP with progressive RNFL and GCL loss or visual field defects may signal concurrent glaucoma and thus warrant the initiation of IOP regulation.33
 |
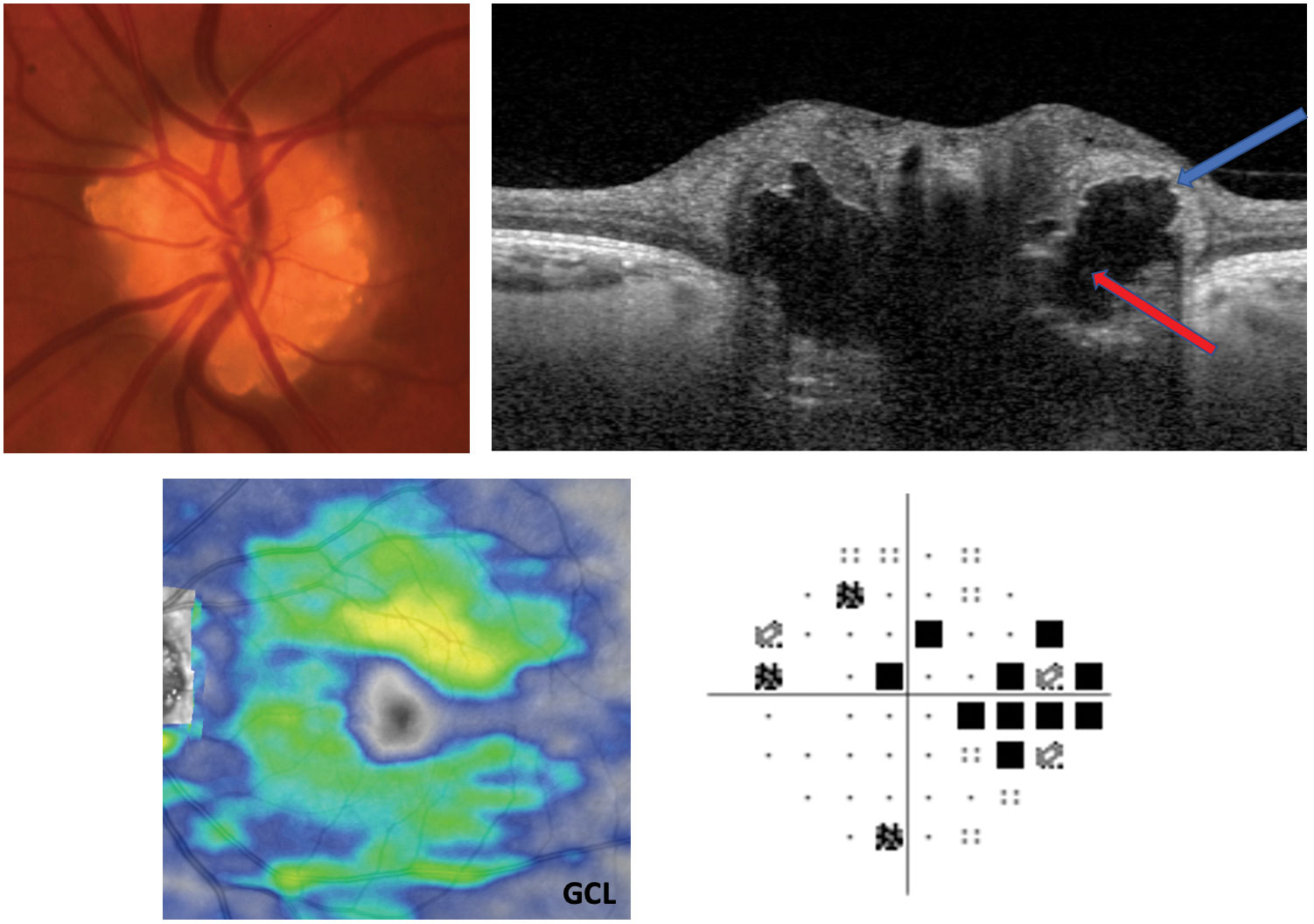
| This patient has advanced glaucoma. Note the posterior and sub-scleral excavation (yellow arrow) of prelaminar tissues (blue arrow) and lamina thinning (red arrow) on vertical OCT cross-section consistent with glaucoma. Advanced GCL loss and corresponding 10-2 grayscale are included. Click image to enlarge. |
Papilledema. True papilledema is bilateral optic disc edema due to increased CSFP and subsequent increased intracranial pressure (ICP). It presents initially with indistinct, hyperemic and elevated rim tissue, potentially with vessel obscuration, hemorrhages, negative spontaneous venous pulsation and Paton’s lines. Papilledema has various systemic etiologies, including compressive brain tumors, venous sinus thrombosis and idiopathic intracranial hypertension (IIH). Confirmation of papilledema from funduscopic and technological examination requires emergent intervention.38
Elevated CSFP in the optic nerve sheath will directly influence laminar position via increased translaminar pressure difference, which produces a net posterior force on the surface of the lamina and a hydrostatic pressure gradient within the prelaminar and laminar tissues. If unmanaged, this mechanical insult will lead to optic nerve swelling from axoplasmic flow stasis, peripapillary expansion and deformation and globe flattening. When ICP is decreased with appropriate treatment (e.g., weight loss, oral acetazolamide, lumbar puncture, shunt surgery), the CSFP within the optic nerve sheath will also decline. Timely management of these cases is critical, as chronic edema can lead to irreversible damage.39
Differentiating papilledema from glaucoma comes into play clinically when managing the long-term effects of papilledema, which include structural optic nerve and ganglion cell atrophy and functional visual decline. Funduscopically, the optic nerve may appear diffusely pale with no excavation or cupping of the rim tissue. If RNFL atrophy is severe (approaching 40µm in thickness), the optic nerve may have overlying damaged astrocytes, which appear similar to glial tissue and may obscure the rim margin.9,31 Post-edema RNFL values vary depending on the chronicity and severity of the papilledema, with some reports of a return to normal ranges and others of mild to drastic atrophy.
OCT can be very helpful in these cases, with certain parameters offering more information than others. One study determined elevated ICP is associated with increased ONH volume and decreased Bruch’s membrane displacement volume. It found that with a decrease in ICP (after lumbar puncture) comes a decrease in ONH volume and in the ganglion cell complex volume, reinforcing how retinal atrophy is also a consequence of optic disc edema. Also of note was the stability of cup volume before and after lumbar puncture, which supports the funduscopic presentation of optic nerve atrophy without cupping.38
Other research examined IIH patients who were treated with acetazolamide or surgical intervention and discovered that ganglion cell loss occurred on average about two months after initial diagnosis, despite treatment, which correlated to the extent of final visual field loss and mean deviation reduction even up to one-year post-treatment. Thus, from a clinical standpoint, although visual field assessment has been considered the gold standard for monitoring functional loss in papilledema, doctors should now consider serial GCA to aid in detection of earlier patterns of neuropathic disease.39
 |
| Pictured here is ONH drusen. Note the hyper-reflective borders (blue arrow) and hyporeflective core of the drusen (red arrow) on EDI-OCT cross-section. A myriad of presentations can occur with both visual fields and GCL. This patient’s GCL is stable, and Humphrey visual fields show a partial arcuate defect, which is a common field result. Click image to enlarge. |
Ischemic optic neuropathies. Anterior ischemic optic neuropathies (AION) initially present with optic disc edema from swelling of the RNFL and are classified as non-arteritic (NAION) or arteritic (AAION) based on presentation and etiology. NAION is the most frequent cause of unilateral optic disc edema in ages 45 to 70 and is characterized by a hyperemic, edematous nerve with flame hemorrhages and arteriolar narrowing that causes sudden, painless vision loss.40
AAION is an ocular emergency which arises from giant cell arteritis, a systemic vasculitis of medium- and large-sized vessels, with variable systemic symptoms such as jaw claudication and temporal headache. AAION presents with a chalky-white, edematous optic disc, with hemorrhages and severe, sudden, painless vision loss. If acute AAION is suspected, emergent intervention is necessary to reduce the risk of progression to bilateral blindness.41
Ischemic optic nerve damage is not governed by IOP-related tissue stress like glaucoma, but instead from a hypoxic state from insufficient posterior ciliary artery circulation, which cascades retinal ganglion cell axonal damage to axonal death. Systemic or nocturnal hypotension and systemic vascular diseases such as diabetes and hypertension may initiate NAION optic nerve non-perfusion.9,40,42
NAION RNFL damage is greatest in superior disc sectors, especially the nasal and temporal sectors. This is likely explained by more tightly packed axons and thus higher oxygen demand, leading to a more rapid hypoxic state in these sectors.43 AAION pathophysiology involves inflammatory occlusion of the short posterior ciliary arteries at the ONH in response to granulomatous vasculitis.41
Regardless of etiology, ischemic insult to the optic nerve will induce pallor and RNFL atrophy without rim tissue excavation or laminar deformation.9 This is clinically observed as “pallor greater than cupping,” distinguishing the aftermath of ischemic insult from glaucoma.42-44 Researchers examined end-stage AION for the presence of cupping and determined 2% of NAION eyes experienced cupping vs. sectoral pallor (72%) or diffuse pallor (28%), while 92% of AAION eyes experienced cupping.43
Optic nerve excavation may be lacking in NAION because the lamina cribrosa is spared from ischemic insult.42 Further topographical investigation with scanning laser ophthalmoscopy shows a difference in ischemic vs. glaucomatous cupping; neither NAION nor AAION experience deep cupping like glaucoma. Research found glaucomatous cup volume was seven times greater than NAION and two times greater than AAION, confirming that while cup size in end-stage AAION and glaucoma may appear similar, it is the glaucomatous nerve experiencing progressive posterior excavation and laminar deformation.44
RNFL edema can vary in severity but is more significant and diffuse in AAION than NAION. As the ischemic insult enters its chronic phase (>six weeks), RNFL edema begins resolving and RNFL atrophy occurs. One study determined that a majority of acute NAION patients experienced superior-quadrant RNFL thickening, with RNFL thinning in chronic NAION predominantly occurring in the superior and temporal quadrants, with significant atrophy progressing for up to six months before plateauing. Compared with glaucoma, this RNFL atrophy is rapid and matches the clinical presentation of defined disc pallor, even approaching OCT “floor” micron values.
Acute and chronic NAION will present with characteristic GCA altitudinal loss, most commonly superior and identifiable before optic disc edema resolution.45 Segmenting out the macular layers shows ganglion cell loss occurring as early as two weeks post-initial presentation, with progressive inner retinal thinning occurring until it levels out at around three months.45 When comparing acute with chronic NAION, the internal plexiform layer (IPL) thinning was the most significant of the macular layers.40,45
This is in direct contrast to glaucoma where less abrupt GCL thinning occurs, IPL thinning is less severe and macular RNFL thinning mimics optic disc RNFL loss.46 Ischemic inner retinal loss corresponds functionally with altitudinal visual field loss, commonly inferior, another hallmark finding of acute and chronic NAION.40,47 Generalized GCA and visual field depression is typically experienced in AAION; be mindful that a patient with severe vision loss >20/200 from either ischemic condition may not have the visual capacity to perform an accurate visual field test.40
Takeaways
A host of non-glaucomatous optic nerve conditions either confound examination or create a clinical appearance of glaucomatous mimicry. Taking a systematic clinical and technological approach, while addressing each patient’s care on an individual basis, will help differentiate optic nerve conditions and reduce patient burden.
Dr. Hogan is a clinical instructor at Southern College of Optometry. Dr. Rixon is an attending optometrist at the Memphis VA, an AAO diplomate in glaucoma and a member of the Optometric Glaucoma Society. Neither author has any financial interests to disclose.
1. Vajaranant TS, Wu S, Torres M, et al. The changing face of primary open-angle glaucoma in the United States: demographic and geographic changes from 2011 to 2050. Am J Ophthalmol. 2012;154(2):303-14.e3. 2. Chandler and Grant’s Glaucoma, Fifth Edition - SLACK BOOKS. Accessed March 28, 2021. https://ebooks.healio.com/product/chandler-grants-glaucoma-fifth-edition. 3. Fraser CL, White AJR, Plant GT, et al. Optic nerve cupping and the neuro-ophthalmologist. Journal Neuroophthalmol. 2013;33(4):377-89. 4. Hayreh SS. Blood supply of the optic nerve head. Ophthalmologica. 1996;210(5):285-95. 5. Susanna R, Vessani RM. New findings in the evaluation of the optic disc in glaucoma diagnosis. Curr Opin Ophthalmol. 2007;18(2):122-128. doi:10.1097/ICU.0b013e328040bfe0 6. Poon LY-C, Solá-Del Valle D, Turalba AV, et al. The ISNT rule: how often does it apply to disc photographs and retinal nerve fiber layer measurements in the normal population? Am J Ophthalmol. 2017;184:19-27. 7. Tezel G. Alterations in the morphology of lamina cribrosa pores in glaucomatous eyes. Br J Ophthalmol. 2004;88(2):251-6. 8. Crawford Downs J, Roberts MD, Sigal IA. Glaucomatous cupping of the lamina cribrosa: A review of the evidence for active progressive remodeling as a mechanism. Exp Eye Res. 2011;93(2):133-40. 9. Burgoyne C. The morphological difference between glaucoma and other optic neuropathies. J Neuroophthalmol. 2015;35 Suppl 1:S8-S21. 10. O’Neill EC, Gurria LU, Pandav SS, et al. Glaucomatous optic neuropathy evaluation project: factors associated with underestimation of glaucoma likelihood. JAMA Ophthalmol. 2014;132(5):560-6. 11. Nixon GJ, Watanabe RK, Sullivan-Mee M, et al. Influence of optic disc size on identifying glaucomatous optic neuropathy. Optom Vis Sci. 2017;94(6):654-63. 12. O’Neill EC, Kong YXG, Connell PP, et al. Gaze behavior among experts and trainees during optic disc examination: does how we look affect what we see? Invest Ophthalmol Vis Sci. 2011;52(7):3976-83. 13. Sanderson J, Rixon A, Williamson J. The ins and outs of pressure gradients. Accessed July 5, 2020. https://www.reviewofoptometry.com/article/the-ins-and-outs-of-pressure-gradients. 14. Quigley HA, Addicks EM, Green WR. Optic nerve damage in human glaucoma. III. Quantitative correlation of nerve fiber loss and visual field defect in glaucoma, ischemic neuropathy, papilledema, and toxic neuropathy. Arch Ophthalmol. 1982;100(1):135-146. 15. Cho H-K, Park JM, Kee C. Effect of optic disc size on correlation between Bruch’s membrane opening-minimum rim width and peripapillary retinal nerve fibre layer thickness. Eye (Lond). 2019;33(12):1930-8. 16. Pilat AV, Gottlob I, Sheth V, et al. Gender- and ethnicity-related differences in optic nerve head topography in healthy indian and caucasian participants. Neuroophthalmology. 2014;38(4):205-12. 17. Turgut B. Pearls for correct assessment of optic disc at glaucoma diagnosis. Published online November 1, 2017. Accessed March 28, 2021. https://www.touchophthalmology.com/glaucoma/journal-articles/pearls-for-correct-assessment-of-optic-disc-at-glaucoma-diagnosis/. 18. Lim CS, O’Brien C, Bolton NM. A simple clinical method to measure the optic disc size in glaucoma. J Glaucoma. 1996;5(4):241-5. 19. Rao HBL, Sekhar GC, Babu GJ, et al. Clinical measurement and categorization of optic disc in glaucoma patients. Indian J Ophthalmol. 2009;57(5):361-4. 20. Wall M, Lee EJ, Wanzek RJ, et al. Temporal wedge defects in glaucoma: structure/function correlation with threshold automated perimetry of the full visual field. J Glaucoma. 2020;29(3):191-7. 21. Bayraktar S, Sultanova G, Cebeci Z, et al. New circumpapillary retinal nerve fiber layer thickness and bruch’s membrane opening-minimum rim width assessment in nonglaucomatous eyes with large discs. J Ophthalmol. 2019;2019:3431217. 22. Hammel N, Belghith A, Bowd C, et al. Rate and pattern of rim area loss in healthy and progressing glaucoma eyes. Ophthalmology. 2016;123(4):760-70. 23. Cazana IM, Böhringer D, Reinhard T, et al. A comparison of optic disc area measured by confocal scanning laser tomography versus Bruch’s membrane opening area measured using optical coherence tomography. BMC Ophthalmol. 2021;21(1):31. 24. Enders P, Adler W, Schaub F, et al. Novel Bruch’s membrane opening minimum rim area equalizes disc size dependency and offers high diagnostic power for glaucoma. Invest Ophthalmol Vis Sci. 2016;57(15):6596-6603. 25. Apple DJ, Rabb MF, Walsh PM. Congenital anomalies of the optic disc. Surv Ophthalmol. 1982;27(1):3-41. 26. Witmer M, Margo C, Drucker M. Tilted optic disks. Survey of Ophthalm. 2010;55(5):403-28. 27. Rebolleda G, Casado A, Munoz-Negrete F. The new Bruch’s membrane opening-minimun rim width classification improves optical coherence tomography specificity in tilted discs. Clinical Ophthalmol. 2016;10:2417-25. 28. Brazitikos PD, Safran AB, Simona F, et al. Threshold perimetry in tilted disc syndrome. Arch Ophthalmol. 1990;108(12):1698-1700. 29. Gürlü VP, Alimgil ML, Benian O. Topographical analysis of the visual field in tilted disk syndrome. Retina. 2002;22(3):366-8. 30. Gili P, Kim-Yeon N, de Manuel-Triantafilo S, et al. Diagnosis of optic nerve head drusen using enhanced depth imaging optical coherence tomography. Eur J Ophthalmol. 2021; doi:10.1177/1120672120986374. 31. Chen JJ, Kardon RH. Avoiding clinical misinterpretation and artifacts of optical coherence tomography analysis of the optic nerve, retinal nerve fiber layer, and ganglion cell layer. J Neuroophthalmol. 2016;36(4):417-38. 32. Tso MO. Pathology and pathogenesis of drusen of the optic nervehead. Ophthalmology. 1981;88(10):1066-80. 33. Allegrini D, Pagano L, Ferrara M, et al. Optic disc drusen: a systematic review: Up-to-date and future perspective. Int Ophthalmol. 2020;40(8):2119-27. 34. Vahlgren J, Malmqvist L, Rueløkke LL, et al. The angioarchitecture of the optic nerve head in patients with optic disc drusen. Neuroophthalmology. 2020;44(1):5-10. 35. Almog Y, Nemet A, Nemet AY. Optic disc drusen demonstrate a hyperechogenic artifact in B mode ultrasound. J Clin Neurosci. 2016;23:111-9. 36. Chang MY, Pineles SL. Optic disk drusen in children. Surv Ophthalmol. 2016;61(6):745-58. 37. Malmqvist L, Wegener M, Sander BA, et al. Peripapillary retinal nerve fiber layer thickness corresponds to drusen location and extent of visual field defects in superficial and buried optic disc drusen. J Neuroophthalmol. 2016;36(1):41-5. 38. Patel MD, Malhotra K, Shirazi Z, et al. Methods for quantifying optic disc volume and peripapillary deflection volume using radial optical coherence tomography scans and association with intracranial pressure. Front Neurol. 2019;10:798. 39. Athappilly G, García-Basterra I, Machado-Miller F, et al. Ganglion cell complex analysis as a potential indicator of early neuronal loss in idiopathic intracranial hypertension. Neuroophthalmology. 2019;43(1):10-7. 40. Ackermann P, Brachert M, Albrecht P, et al. Alterations of the outer retina in non-arteritic anterior ischaemic optic neuropathy detected using spectral-domain optical coherence tomography. Clin Exp Ophthalmol. 2017;45(5):496-508. 41. Tian G, Chen W, Chen Q, et al. Giant cell arteritis presenting as bilateral anterior ischemic optic neuropathy: a biopsy-proven case report in Chinese patient. BMC Ophthalmol. 2018;18(1):282. 42. Punjabi OS, Tanna AP, Rosenberg MA. Optic disk excavation in nonarteritic anterior ischemic optic neuropathy. J Glaucoma. 2011;20(2):71-3. 43. Danesh-Meyer HV, Savino PJ, Sergott RC. The prevalence of cupping in end-stage arteritic and nonarteritic anterior ischemic optic neuropathy. Ophthalmology. 2001;108(3):593-8. 44. Danesh-Meyer HV, Boland MV, Savino PJ, et al. Optic disc morphology in open-angle glaucoma compared with anterior ischemic optic neuropathies. Invest Ophthalmol Vis Sci. 2010;51(4):2003-10. 45. Akbari M, Abdi P, Fard MA, et al. Retinal ganglion cell loss precedes retinal nerve fiber thinning in nonarteritic anterior ischemic optic neuropathy. J Neuroophthalmol. 2016;36:141-6. 46. Fard MA, Afzali M, Abdi P, et al. Comparison of the pattern of macular ganglion cell-inner plexiform layer defect between ischemic optic neuropathy and open-angle glaucoma. Invest Ophthalmol Vis Sci. 2016;57(11):1011-6. 47. Erlich-Malona N, Mendoza-Santiesteban CE, Hedges TR, et al. Distinguishing ischaemic optic neuropathy from optic neuritis by ganglion cell analysis. Acta Ophthalmol. 2016;94(8):e721-6. |
