The Real-World Cataract Evaluation
For better post-op visual function and quality of life, focus on things that matter to patients.
By Jacqueline Theis, OD

Release Date: June 15, 2019
Expiration Date: June 15, 2022
Estimated Time to Complete Activity: 2 hours
Jointly provided by Postgraduate Institute for Medicine (PIM) and Review Education Group
Educational Objectives: After completing this activity, the participant should be better able to:
- Individualize the cataract evaluation based on the patient’s particular needs and routines.
- Recognize the real-life vision test (or similar objective real-life visual function testing) as a potential adjunctive means of cataract evaluation.
- Identify underlying diseases that increase cataract risk and affect post-op cataract outcomes.
- Discuss IOL choices based not only on clinical measures, but also on patient’s subjective needs and preferences as well as real-life vision testing results.
- Describe the benefits and risks of simultaneous bilateral cataract surgery.
Target Audience: This activity is intended for optometrists engaged in the care of patients with cataracts.
Accreditation Statement: In support of improving patient care, this activity has been planned and implemented by the Postgraduate Institute for Medicine and Review Education Group. Postgraduate Institute for Medicine is jointly accredited by the Accreditation Council for Continuing Medical Education, the Accreditation Council for Pharmacy Education, and the American Nurses Credentialing Center, to provide continuing education for the healthcare team. Postgraduate Institute for Medicine is accredited by COPE to provide continuing education to optometrists.
Faculty/Editorial Board: Jacqueline Theis, OD.
Credit Statement: This course is COPE approved for 2 hours of CE credit. Course ID is 62686-AS. Check with your local state licensing board to see if this counts toward your CE requirement for relicensure.
Disclosure Statements:
Dr. Theis consults for C. Light Technologies.
Managers and Editorial Staff: The PIM planners and managers have nothing to disclose. The Review Education Group planners, managers and editorial staff have nothing to disclose.
Optometrists are often the first to detect, diagnose and counsel patients regarding cataracts, and we must take this responsibility seriously. Cataracts are still the leading cause of vision loss in the United States.1 By 2020, an estimated 30.1 million Americans will have cataracts.1
In spite of the high success rate of cataract surgery, it is not without risk, so we must ensure that the benefits outweigh the risks of complications for each patient. Cataract evaluation requires an individualized approach beyond measuring visual acuity and performing a slit-lamp examination. We must understand how a patient uses their vision in their daily life. We need to know what type of cataract they have and, when combined with patient demographics, what this may indicate about their general health. Finally, we need to set appropriate patient expectations for pre- and postoperative care.
To do this, we need to bring the real world into the exam room and consider each patient’s visual needs and experiences as they relate to the objective exam findings.
Individualized Evaluation
The lens of the eye is an anatomically simple yet metabolically complicated structure composed of four nuclei, a cortex and a capsule. The biochemical properties of the lens allow for its continued growth while maintaining transparent functionality, but also makes the lens vulnerable to opacification.
Technically, a cataract is any opacity in the lens—whether a small focal opacity or a diffuse loss of transparency. Cataracts become clinically significant when the patient has a significant reduction in visual acuity, a noticeable functional impairment in their life or both.
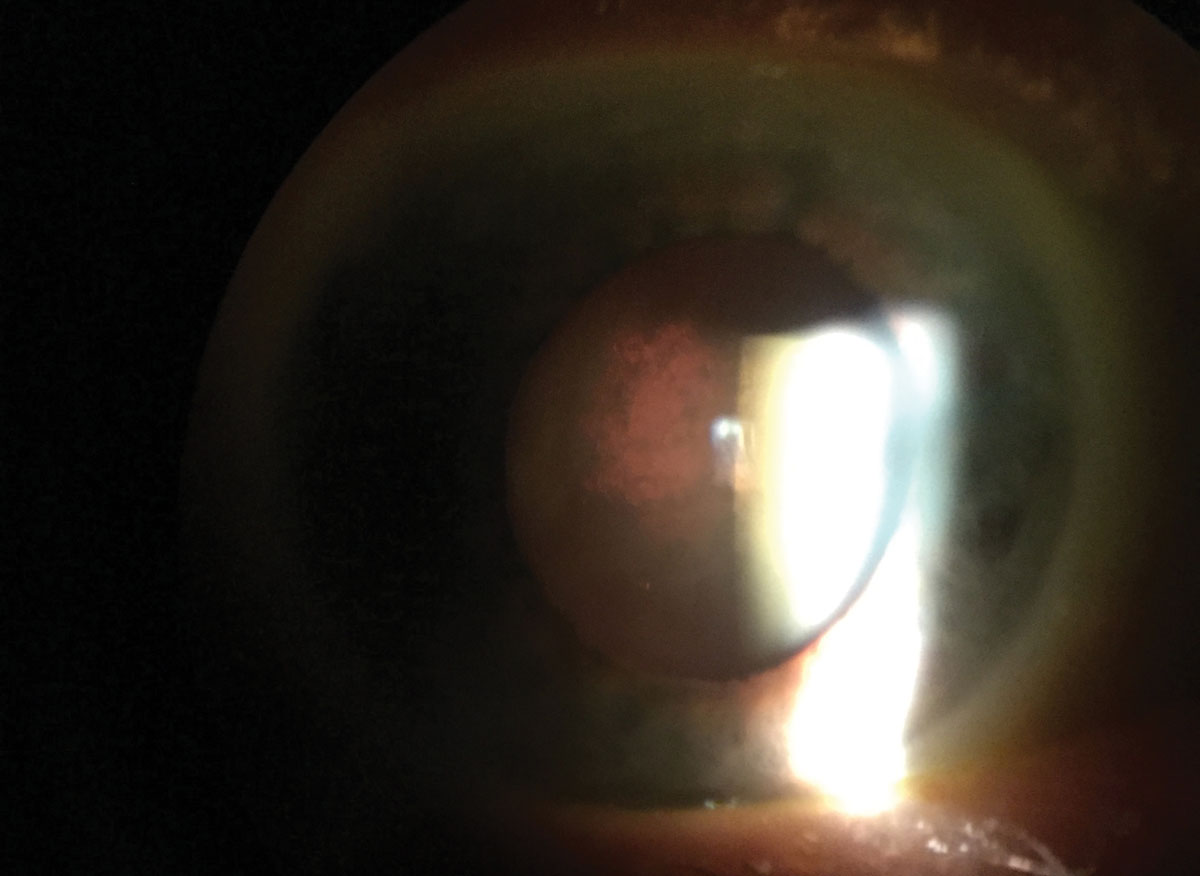 |
| Posterior subcapsular cataracts, such as this one in retroillumunation, are not always part of the nornal aging process. Photo: Marcellus Chow, OD. Click image to enlarge. |
When a patient presents to your office with cataract complaints, how should you begin your evaluation?
Case history. For cataract evaluation, the case history is critical. Every person is unique in his or her occupation, hobbies, responsibilities and visual demands. Getting to know how your patient uses their eyes in their day-to-day life will uncover how their reduced vision will impact their needs and routines, and whether the pursuit of surgery is indicated.
Additionally, taking time to establish a detailed case history helps foster the doctor-patient relationship, which is essential as the patient needs to trust that the recommendations regarding their cataract management is based on their individual needs.
Visual acuity. A variety of visual symptoms related to cataract development may bring patients into your office. The most obvious is reduced visual acuity, with complaints such as, “Things just aren’t as clear as they used to be,” “My vision seems blurry,” “I can’t read small font,” or “I have to get very close to road signs before I can read them.” These common visual acuity complaints are easily quantified by measuring the patient’s best-corrected visual acuity (BCVA) for each eye and both eyes open using standard distance acuity charts and near reading cards.
Surgical intervention is usually indicated when a patient’s BCVA is 20/40 or worse, as this level of vision may disqualify them from driving. Studies show that physical and legal impediments to driving can lead to loss of independence, which may increase rates of depression.2,3 However, some patients may not be the driver in their household and would prefer to delay surgery until their vision is further reduced and interferes with their everyday activities. It’s important to ask patients with reduced BCVA if their vision makes them feel unsafe or prohibits them from pursuing other activities they enjoy beyond driving.
It’s always the patient’s choice to pursue or delay surgery. It’s our job to educate them on the benefits and possible complications of pursuing or delaying cataract surgery, as well as inform them of what and how their reduced visual function may impact their lifestyle. Patients need to know that cataracts will continue to grow and cause gradual decreased vision with time. If a patient prefers to delay surgery, we must educate the patient on the risks of increasing vision loss.
For instance, patients with ambulatory and mobility issues or degenerative diseases such as Parkinson’s must understand that their reduced vision may further compromise their health and safety, as reduced vision will increase their risk of falling.
Additionally, if the patient waits too long for surgery, their risk of postoperative complications increases. Late-stage cataracts are more optically dense and may require more phacoemulsification energy, which can cause greater corneal edema and endothelial cell loss.2,3 Providers should follow up with patients who delay cataract surgery to determine if functional visual disability develops.
Patients with BCVA better than 20/40 who feel their vision prohibits them from functioning in their activities of daily living can be more complicated to manage. While high-contrast threshold acuity of individual letters read at a distance is documented at every eye examination, this does not always translate to real-world visual tasks, including reading phrases and sentences. To objectively assess near vision complaints, a variety of reading cards are available—in high and low contrast editions, with letters, words, phrases, and sentences—to better understand how the patient’s reduced vision impacts their reading ability.
If the patient is an avid reader and feels their vision inhibits their functional reading, ask the patient what they are having trouble reading and where they like to read it. We can’t assume that everyone with a reading complaint is struggling to read 8pt (1M) standard newspaper print, which requires a visual acuity of 20/40. Many books, like small print Bibles, may use 5pt (0.6M) print. Medicine bottle labels that describe side effects and dosage information often use even smaller 3pt (0.4M) print, requiring 20/20 acuity for resolution. Ask these patients to bring their reading material with them to the examination so you can understand their visual acuity demands for their occupation and lifestyle.
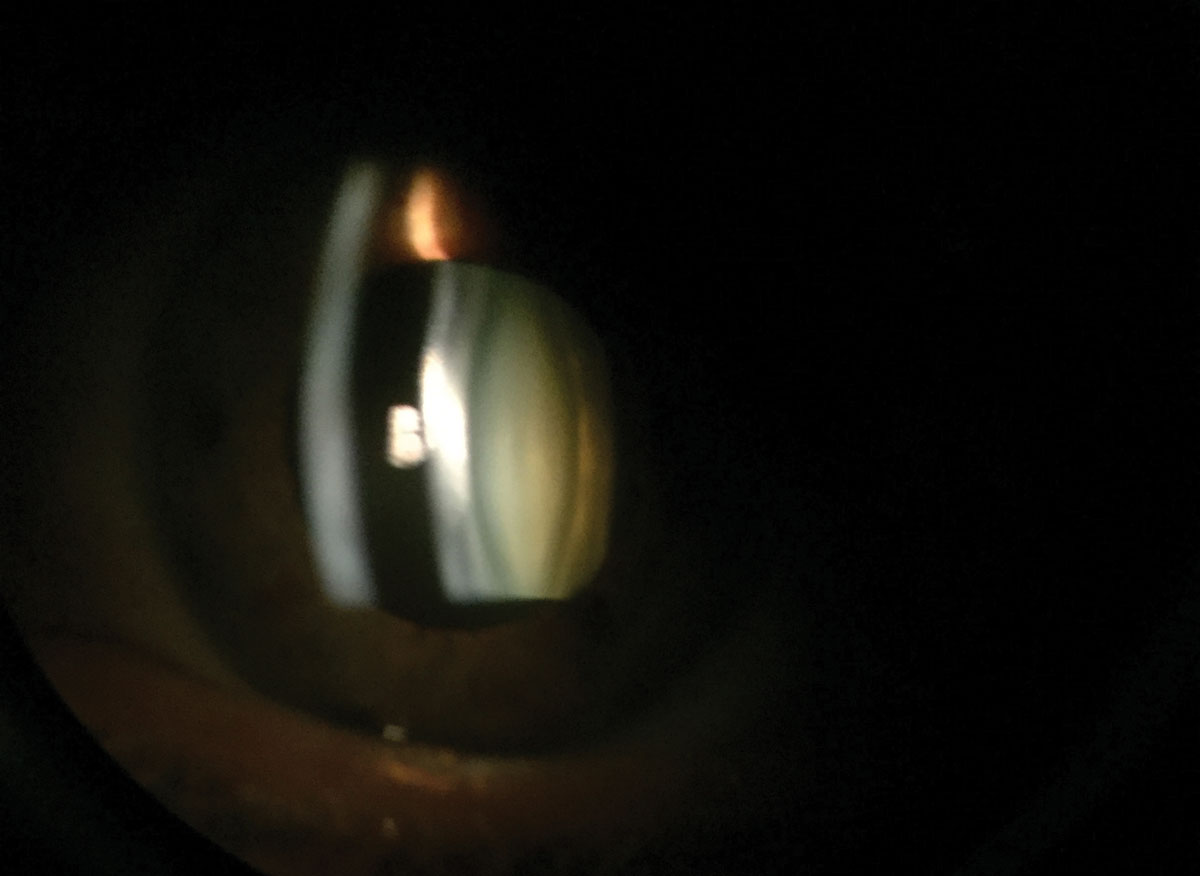 |
| Parallelepiped slit lamp examination reveals a nuclear sclerotic cataract. Photo: Marcellus Chow, OD. Click image to enlarge. |
While the patient may be able to adapt their lifestyle to their reduced vision using magnifiers, increased lighting or increased font size, some individuals may be unable to use these adaptations due to dexterity issues or visual acuity demands outside of their control. In these cases, earlier cataract surgery referral may be appropriate.
A patient may be an appropriate candidate for cataract surgery if they cannot read comfortably for prolonged periods of time at their threshold visual acuity. It is commonly recognized clinically that comfortable reading for prolonged periods of time requires a font size that is larger than the individual’s threshold acuity. In these cases, primary eye care providers need to go beyond the standard examination to objectively identify and document the level of debilitation the patient’s cataracts have on their lifestyle to help advocate for surgical intervention if the patient so desires.
Look Beyond Visual Acuity
Cataracts may impact aspects of visual function beyond visual acuity, including contrast sensitivity, glare and color vision.
Contrast sensitivity. This is one of the most important real-world-related visual functions we can test, as it relates directly to the quality of vision in variable lighting conditions. A patient with reduced contrast sensitivity may report: “Concrete stairs are more difficult to navigate,” “I can’t see in low light or on rainy days,” “I have trouble recognizing faces in crowds,” or “I feel unsafe driving under changing lighting conditions, especially tree coverage, tunnels and bridges.”
Contrast can be measured directly using a variety of objective charts including, but not limited, to: the Pelli-Robson, VisTech and Mars Letter contrast sensitivity tests.
Glare. A patient with cataracts may also report having “problems with glare.” This is a difficult complaint to address because it can stem from many different diagnoses, including ocular surface disease, corneal edema, cataracts and uncorrected astigmatism, to name a few—many of which may coexist in a single patient.
Patients need to elaborate further on their glare complaints and describe which lighting or environmental conditions provoke the most glare, along with the time of day and the frequency they encounter glare throughout the day, as this information will guide clinical insight into the etiology of the glare complaint and even the type of cataract.
We have many ways to objectively measure glare. One easy tool is to measure the patient’s ambient light or brightness acuity (i.e., their visual acuity when ambient overhead exam room lighting is on, or when a penlight is directed toward the patient while they read the eye chart). A drastic reduction in visual acuity when confronted with glare objectively supports a patient’s complaints of debilitating glare and would bolster the need for a referral for cataract surgery.
Color vision. While the subtle changes in color vision that occur with nuclear sclerotic cataracts are not always apparent to every patient, there are many patients for whom this visual change can be life-altering. Artists or interior designers, who make a living based upon their color vision, may be debilitated by these subtle changes, and further exploration using a specialized color vision test, such as the 100-hue color test, may help objectively document their subjective complaints.
At present, primary eye care providers have a limited capacity to objectively document a patient’s visual experience and the impact of cataracts on subjective visual function because the optometrist’s exam lane does not always mimic real-world conditions. We often have to compromise by using alternate examination techniques such as contrast sensitivity, color vision and artificial glare testing to quantify the patient’s subjective visual disability.
| What is the Real-Life Vision Test (RLVT)? The RLVT is a performance-based measure for assessing functional vision. It was developed in recent years by researchers in China, who showed that it could reveal additional information than what standard clinical measures or subjective surveys can provide, particularly in cataract patients.4,5 The RLVT is a standardized assessment that includes several real-world functional visual tasks, such as identifying particular letter characters in newsprint, matching and threading buttons, recognizing street signs from a distance, and others. “RLVT can help the doctors to examine the patient as a ‘whole person’ with the fact that it could measure visual disability directly by assessing what a person ‘can see’ in real life. In such way, RLVT may facilitate a more thorough evaluation of changes in functional outcomes and the efficacy of cataract surgery,” the researchers concluded in their study.4 |
There is a need for real-life visual function testing, such as the real-life vision test, to be clinically accessible to help providers evaluate and manage cataracts more appropriately.4,5
Lens Evaluation
After adequately documenting the nature and severity of the patient’s visual impairment, you need to complete a comprehensive dilated eye examination to evaluate the lens and rule out coexisting ocular and systemic disease.
The type of cataract that forms and its specific anatomical location impacts the optics of the lens differently. As such, each patient’s symptoms will depend on the pathophysiological properties and type of cataract formed within the individual’s lens. When evaluating the lens, identify the location, type and severity of opacification.
Nuclear cataract. An opacity in the fetal or embryonic nucleus is classified as a nuclear cataract. The most common type, senile nuclear sclerosis, appears as a brunescence of the inner lens that occurs due to lens protein fiber modification by oxidation, proteolysis and glycation, which eventually leads to protein aggregation into high molecular weight particles.
These high molecular weight particles scatter light and will, over time, increase the optical density of the nuclei, leading to a myopic shift in the refractive properties of the lens. Additionally, urochrome pigment concentration in lens fibers leads to a yellowing of the lens.6 Patients with nuclear sclerotic cataracts often complain of color vision changes, glare, monocular diplopia and reduced vision under night and low-light conditions.7
Cortical cataract. These evolve from an electrolyte imbalance within the cortex, causing overhydration and liquification of the lens fibers. Eventually, this overhydration causes the formation of vacuoles, clefts, wedges, lamellar separations or a combination of all of these.6 The anterior or posterior location of cortical cataracts signifies the localized anatomical breakdown. Patients with cortical cataracts often complain of monocular diplopia and reduced vision that is worse under bright lights and glare conditions, especially when the cataracts extend into the pupillary region of the lens.7
Posterior subcapsular cataract (PSCs). Unlike nuclear sclerotic and cortical cataracts, these are not always a normal part of the aging process, and the exact causative mechanism is unknown.8 PSCs develop due to loss of lens fiber nuclei in the posterior pole. In response to this loss, epithelial cells aberrantly migrate to replace the nuclei. However, these epithelial cells cluster and form balloon cells, which interdigitate with adjacent lens fibers and deeper cortical lens fibers, breaking them down. This clustering and breakdown leads to a lacy, granular opacification of the lens that is unfortunately located in an inherently important optical region of the lens.
 |
| Click table to enlarge. |
Even small PSCs can be visually devastating to patients, causing them to have reduced vision that is worse in bright light or miotic conditions such as bright sunlight, oncoming headlight traffic at night or near visual tasks.7
While we all like to blame aging for cataract development, there is no single process that causes lens opacification in a patient. A variety of precipitating risk factors exists, including nutrition, ultraviolet light exposure, genetics, trauma, inflammation, high myopia, acute angle closure, systemic disease and certain medications (Table 1).7,9
The type of cataract a patient has in relation to their demographics may be an indicator of an underlying systemic disease, such as diabetes or atopic dermatitis, that is undiagnosed and warrants appropriate referral (Table 2).7,10
During the preoperative dilated slit-lamp examination, be sure to closely examine for any pseudoexfoliative deposition on the corneal endothelium, anterior lens capsule, lens zonules, iris and trabecular meshwork. Pseudoexfoliation puts the patient at a preoperative risk for abnormal anterior chamber depth, small pupillary dilation due to iridodenesis, lens instability (phacodenesis), subluxation due to zonulopathy and glaucoma.
Additionally, these patients are at higher intra- and postoperative risk for complications such as endothelial cell damage, cystoid macular edema, intraocular pressure (IOP) spikes and uveitis, as well as lens dislocation due to zonular dehiscence.11
Comorbidities and Complications
Patients with concomitant cataracts and glaucoma can be challenging to manage. Cataract surgery may lower IOP in some patients, but may cause IOP elevation/fluctuation in other patients intra- and postoperatively.12 You and your patient need to weigh the possible risks of glaucoma exacerbation with the benefits of improved visual acuity, contrast sensitivity and quality of life post-cataract surgery prior to referral.13
Preoperative corneal disease and anterior segment findings should be identified and proactively managed, as they can impact postoperative outcomes. Anterior segment findings that may predispose the patient to postoperative endophthalmitis include dacryocystitis, blepharitis, chronic conjunctivitis, lagophthalmos, ectropion, entropion and ocular surface disease. Appropriate management of these conditions can reduce infection risk and improve visual outcomes.14
While rare, cataract surgery can cause recurrence of herpes simplex keratitis in patients with preexisting episodes.15 Prophylactic antiviral treatment may be indicated pre- and postoperatively to reduce this risk.
Patients with corneal stromal dystrophies or scarring may require referral to a corneal specialist who can perform a simultaneous keratoplasty. In addition, those diagnosed with guttata or Fuchs’ corneal dystrophy may require additional pre-op education, as cataract surgery can further endothelial cell loss, and guttata may increase the risk of prolonged postoperative corneal edema.16
Educating patients preoperatively about their individual corneal and anterior segment disease, and what they can expect after recovery, will provide them with a better subjective postoperative experience.
While cataract surgery is low-risk, complications may still arise during or after surgery, including retinal detachment, exacerbation of glaucoma, cystoid macular edema (CME) and, rarely, infection. Patients who are younger, have high myopia, are male or have a posterior capsular rupture during their surgery are at a higher risk for developing a rhegmatogenous retinal detachment after cataract surgery.17
The most frequent cause of poor visual outcome after uneventful cataract surgery is postoperative CME. Patients with preexisting hypertension, diabetes, capsule rupture, epiretinal membrane, uveitis, retinal vein occlusion or retinal detachment repair are at higher risk for this postoperative complication.18-20
To appropriately set postoperative vision expectations, preoperative potential acuity testing is recommended for any patient with coexisting ocular disease. This can be done using potential acuity pinhole (PAP) or pinhole acuity meter (PAM) testing. If a patient with a cataract has PAP or PAM testing worse than 20/20, clinicians should further evaluate for comorbid etiologies such as corneal, retinal or optic nerve disease, in addition to the cataract that may be causing reduced vision.
 |
| Click table to enlarge. |
IOL Selection
Once you and your patient have decided the benefits of cataract surgery outweigh the risks, you need to discuss the IOL options based on clinical measures, as well as the patient’s subjective needs and preferences.
Lifestyle-specific questions are imperative in guiding the patient in IOL decisions. Patients often feel that they need to purchase the most expensive option to have the best visual outcome. This misconception may lead a patient to choose a premium IOL that is not in their best interest, leading to postoperative visual complaints.
The key to individualized IOL selection is understanding the patient’s postoperative goals relating to how they want to use their eyes, how glasses-dependent they prefer to be, and if they have the ability to adapt to some of the inherent imperfections of the lens options. For example, some patients might like the idea of monovision at first. But after a contact lens trial, they realize that they are unable to adapt to this modality and decide not to pursue this IOL option.
Just as there are many types of progressive lenses, there are many types of premium, non-monofocal IOLs, including accommodating, multifocal and extended depth-of-focus IOLs—any of which may be appropriate for an individual’s needs. So, for patients who want to be free of glasses and see all distances comfortably, a premium IOL might be the answer.
However, due to the high optical demands of premium lenses, patients who are highly visually sensitive, have precise visual demands, have moderate to high astigmatism that requires toric correction, have macular pathology or are at risk for zonular dehiscence are poorer candidates for these lenses. Patients with moderate to high corneal astigmatism who want to be less dependent on distance glasses would benefit from monofocal toric IOLs.
To adequately guide your patient, collaborate with your local ophthalmologist to understand the specific IOL options they provide in their office, what the appropriate patient expectations should be for each lens, the types of surgical equipment they use and the procedures they offer.
Cataract Procedures
Phacoemulsification with a femtosecond laser has different risks than the manual ultrasound procedure. Additionally, while the majority of cataract surgeons offer delayed sequential bilateral cataract surgery (DSBCS)—where the patient has one eye operated on, followed by the second eye on a later date—some surgeons now offer immediately sequential bilateral cataract surgery (ISBCS), where the patient has both eye operations on the same day.21 ISBCS has the ability to increase surgical efficiency, which can increase patient access to surgery by reducing wait time and cost to both the patient and healthcare payer.22
In patients who are adequate candidates for ICBCS (i.e., low risk for postoperative complications and have home/mobility support needed for postoperative care), recent studies show equivalent postoperative visual outcomes and patient satisfaction without increased complication risk.23 However, patients at higher risk for postoperative complications, such as high myopes or patients with diabetes or corneal guttata, may risk bilateral complications, in which case they would be a better candidate for DSBCS.
Many barriers to accessing surgical treatment exist, including insurance, treatment costs, surgical availability and patient awareness. We need to advocate for our patients to help break down these barriers.
As the number of patients with cataracts continues to rise, and the life expectancy and inherent visual demands increase, optometrists need to individualize their cataract evaluation and management to align with the needs of the patient. A thorough understanding of the patient’s lifestyle and functional visual needs will help the optometrist guide the patient in deciding when to pursue cataract surgery and which surgical options will enable the patient to visually pursue and enjoy their life.
Dr. Theis is a staff optometrist at Kaiser Permanente in San Rafael, Calif.
|
1. Centers for Disease Control and Prevention. Vision Health Initiative: Common Eye Disorders. September 2015. www.cdc.gov/visionhealth/basics/ced. 2. Gimbel HV, Dardzhikova AA. Consequences of waiting for cataract surgery. Curr Opin Ophthalmol. 2011;22(1):28-30. 3. Hodge W, Horsley T, Albiani D, et al. The consequences of waiting for cataract surgery: a systemic review. CMAJ. 2007;176(9):1285-90. 4. Ni W, Li X, Hou Z, et al. Impact of cataract surgery on vision-related life performances: the usefulness of Real-Life Vision Test for cataract surgery outcomes evaluation. Eye. 2015;29(12):1545-54. 5. Ni W, Li X, Ao M, et al. Using the real-life vision test to assess the functional vision of age-related cataract patients. Eye. 2012;26(11):1402-11. 6. Datiles MB, Kinoshita JH. Pathogenesis of Cataracts. In: Tasman W, Jaeger EA, eds. Duane’s Clinical Ophthalmology, Vol 1. Philadelphia: JB Lippincott; 1991:1-14. 7. Kanski JJ. Ch. 12–Lens. In: Clinical Ophthalmology: A Systematic Approach, 6th ed. Edinburgh: Butterworth Heineman Elsevier; 2007. 8. Remington LA. Ch. 5–Crystalline Lens. Clinical Anatomy of the Visual System, 2nd ed. St Louis: Elsevier; 2005. 9. Hutnik CM, Nichols BD. Cataracts in systemic diseases and syndromes. Curr Opin Ophthalmol. 1999:10(1):22-28. 10. Bagheri N, Wajda BN. Ch. 13–General Ophthalmic Problems. In: The Wills Eye Manual, 7th ed. Philadelphia: Wolters Kluwer; 2017. 11. Fontana L, Coassin M, Lovieno A, et al. Cataract surgery in patients with pseudoexfoliation syndrome: current updates. Clin Ophthalmol. 2017 Jul;11:1377-83. 12. Law SK, Riddle J. Management of cataracts in patients with glaucoma. Int Ophthalmol Clin. 2011;51(3):1-18. 13. Xu X, Sun Q. Vision-related quality of life outcomes of cataract surgery in advanced glaucoma patients. J Glaucoma. 2016;25(1):e5-11. 14. Movahedan A, Djalilian AR. Cataract surgery in the face of ocular surface disease. Curr Opin Ophthalmol. 2012;23(1):68-72. 15. Barequet IS, Wasserzug Y. Herpes simplex keratitis after cataract surgery. Cornea. 2007;26(5):615-7. 16. Greene JB, Mian SI. Cataract surgery in patients with corneal disease. Curr Opin Ophthalmol. 2013;24(1):9-14. 17. Haug SJ, RB Bhisitkul. Risk factors for retinal detachment following cataract surgery. Curr Opin Ophthalmol. 2012;23(1):7-11. 18. Yonekawa Y, Kim IK. Pseudophakic cystoid macular edema. Curr Opin Ophthalmol. 2012;23(1):26-32. 19. Chu CJ, Johnston RL, Buscombe C. Risk factors and incidence of macular edema after cataract surgery: a database study of 81984 eyes. Ophthalmology. 2016;123(2):316-23. 20. Boscia F, Giancipoli E, D’ Amico Ricci G, Pinna A. Management of macular oedema in diabetic patients undergoing cataract surgery. Curr Opin Ophthalmol. 2017;28(1):23-28. 21. Popovic M, Campos-Moller X, Schlenker MB, Ahmed IIK. Efficacy and safety of femtosecond laser-assisted cataract surgery compared with manual cataract surgery. Ophthalmology. 2016;123(10):2113-26. 22. Singh R, Dohlman TH, Sun G. Immediately sequential bilateral cataract surgery: advantages and disadvantages. Curr Opin Ophthalmol. 2017;28(1):81-86. 23. Herrington LJ, Liu L, Alexeff S, et al. Immediate sequential vs. delayed sequential bilateral cataract surgery: Retrospective comparison of postoperative visual outcomes. Ophthalmology. 2017;124(8):1126-35. |
