 |
Staging of Diabetes and Diabetic Retinopathy
Recognizing the role of the OD throughout this process is critical.
By Josh Z. Yuen, OD, PhD
Release Date: June 15, 2022
Expiration Date: June 15, 2025
Estimated Time to Complete Activity: 2 hours
Jointly provided by Postgraduate Institute for Medicine (PIM) and Review Education Group
Educational Objectives: After completing this activity, the participant should be better able to:
- Explain the pathophysiology and staging of diabetes.
- Determine their role in the management of these patients at each stage.
- Comanage diabetic retinopathy patients effectively.
- Communicate with patients about diabetes and diabetic retinopathy.
Target Audience: This activity is intended for optometrists engaged in managing patients with diabetes.
Accreditation Statement: In support of improving patient care, this activity has been planned and implemented by PIM and the Review Education Group. PIM is jointly accredited by the Accreditation Council for Continuing Medical Education, the Accreditation Council for Pharmacy Education and the American Nurses Credentialing Center to provide CE for the healthcare team. PIM is accredited by COPE to provide CE to optometrists.
Reviewed by: Salus University, Elkins Park, PA
Faculty/Editorial Board: Josh Z. Yuen, OD, PhD
Credit Statement: This course is COPE approved for 2 hours of CE credit. Activity #124080 and course ID 78771-SD. Check with your local state licensing board to see if this counts toward your CE requirement for relicensure.
Disclosure Statements: PIM requires faculty, planners and others in control of educational content to disclose all their financial relationships with ineligible companies. All identified conflicts of interest are thoroughly vetted and mitigated according to PIM policy. PIM is committed to providing its learners with high-quality, accredited CE activities and related materials that promote improvements or quality in healthcare and not a specific proprietary business interest of an ineligible company.
Those involved reported the following relevant financial relationships with ineligible entities related to the educational content of this CE activity: Author: Dr. Yuen has no relevant financial interests to disclose. Managers and Editorial Staff: The PIM planners and managers have nothing to disclose. The Review Education Group planners, managers and editorial staff have nothing to disclose.
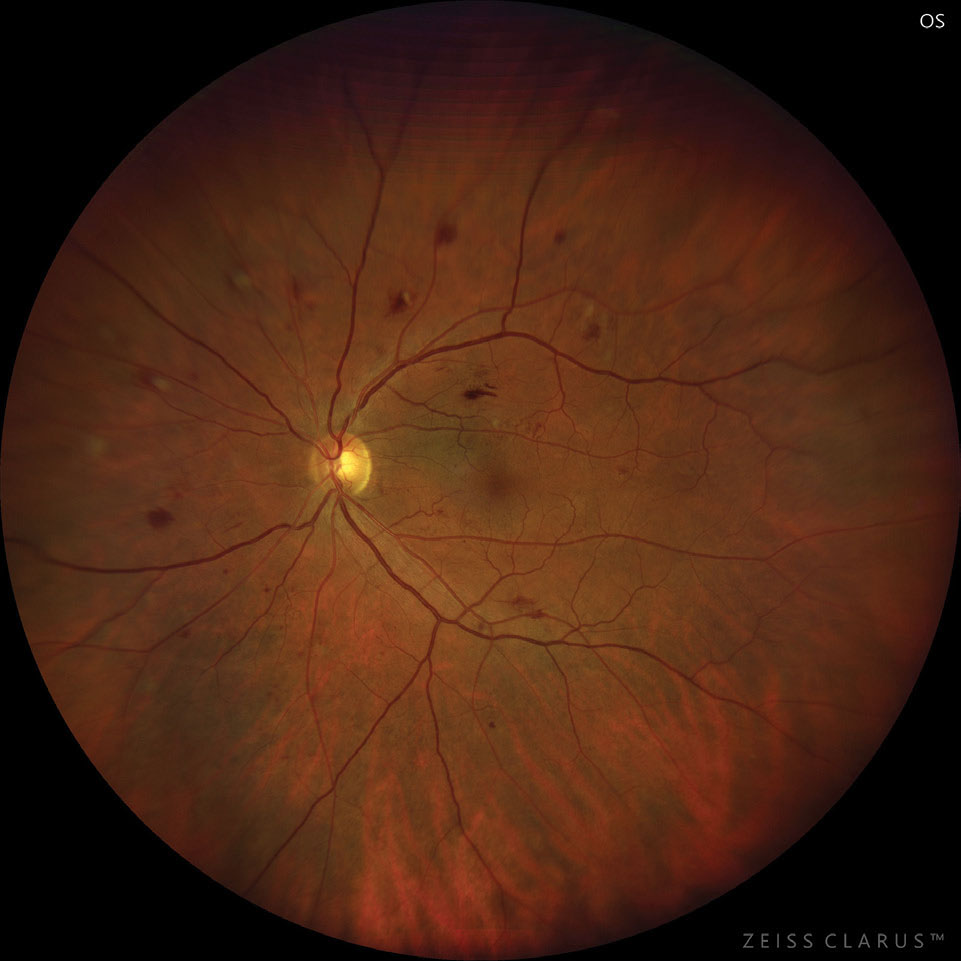 |
| Moderate NPDR care consists of several management techniques, as there is currently no treatment. Photo: Jay M. Haynie, OD. Click image to enlarge. |
Based on the 2022 National Diabetes Statistics report from the CDC, 37 million people in the United States have diabetes mellitus (DM). This number is expected to increase to 55 million by 2030. Diabetes is the leading cause of lower-limb amputations and kidney failure. It is also the leading cause of blindness in working-age adults. Additionally, 11.7% of US adults with diabetes had vision disability, including blindness, in a survey conducted in 2018. Nearly half of patients with diabetes will have diabetic retinopathy (DR) by 2030.1,2
As first-line primary eyecare providers, optometrists play an important role in recognizing DR earlier on, educating patients on the risk factors for diabetes and DR, evaluating the staging of DR and deciding how often to follow-up with patients, as well as when to refer to a retinal specialist.
This article will provide an overview of diabetes, explain the pathophysiology/staging of DR and offer advice regarding what ODs should know, say and do at each stage of the disease.
Types of Diabetes
There are three major types of diabetes: type 1, type 2 and gestational, and it is important that ODs have a clear picture of each.3
Type 1 DM. This is typically, but not always, diagnosed before the age of 30 and was formerly known as juvenile-onset or insulin-dependent diabetes. This type of diabetes accounts for 5% to 10% of the DM population. The mechanism of type 1 DM is the destruction of pancreatic beta-cells (autoimmune reaction) to the degree that ultimately no endogenous insulin is produced.
Type 2 DM. This is the major diabetes type in the US population (90% to 95%). It is typically, but not always, adult-onset, might be without symptoms and usually is characterized by slow progression. Its mechanisms include reduced pancreatic beta-cell insulin secretion, overlaid on top of insulin resistance. Insulin-sensitive tissues become unable to respond to insulin appropriately.4
Gestational DM. This very common subtype of diabetes occurs in 2% to 10% of pregnancies, per the CDC. Gestational diabetes typically resolves by itself after delivery, but both the mother and baby have a higher chance of developing type 2 diabetes in the future (up to 50% maternal risk).
Prediabetes. Based on a CDC report, there are 96 million people in the United States (around 30% of the country’s population) with prediabetes. This is defined as glucose levels higher than normal but not high enough to reach the numeric threshold for diagnosis of diabetes. The pathology includes insulin resistance or beta-cell failure to compensate, just as in type 2 diabetes. These patients may have impaired glucose tolerance, impaired fasting glucose or glycosylated hemoglobin (A1c) levels above normal (5.7% to 6.4%). Fortunately, prediabetes often can be reversed or its progression to type 2 DM slowed by lifestyle modification.5 ODs should encourage patients to work with their primary care providers (PCP) in order to prevent the possible progression of prediabetes into type 2 diabetes.
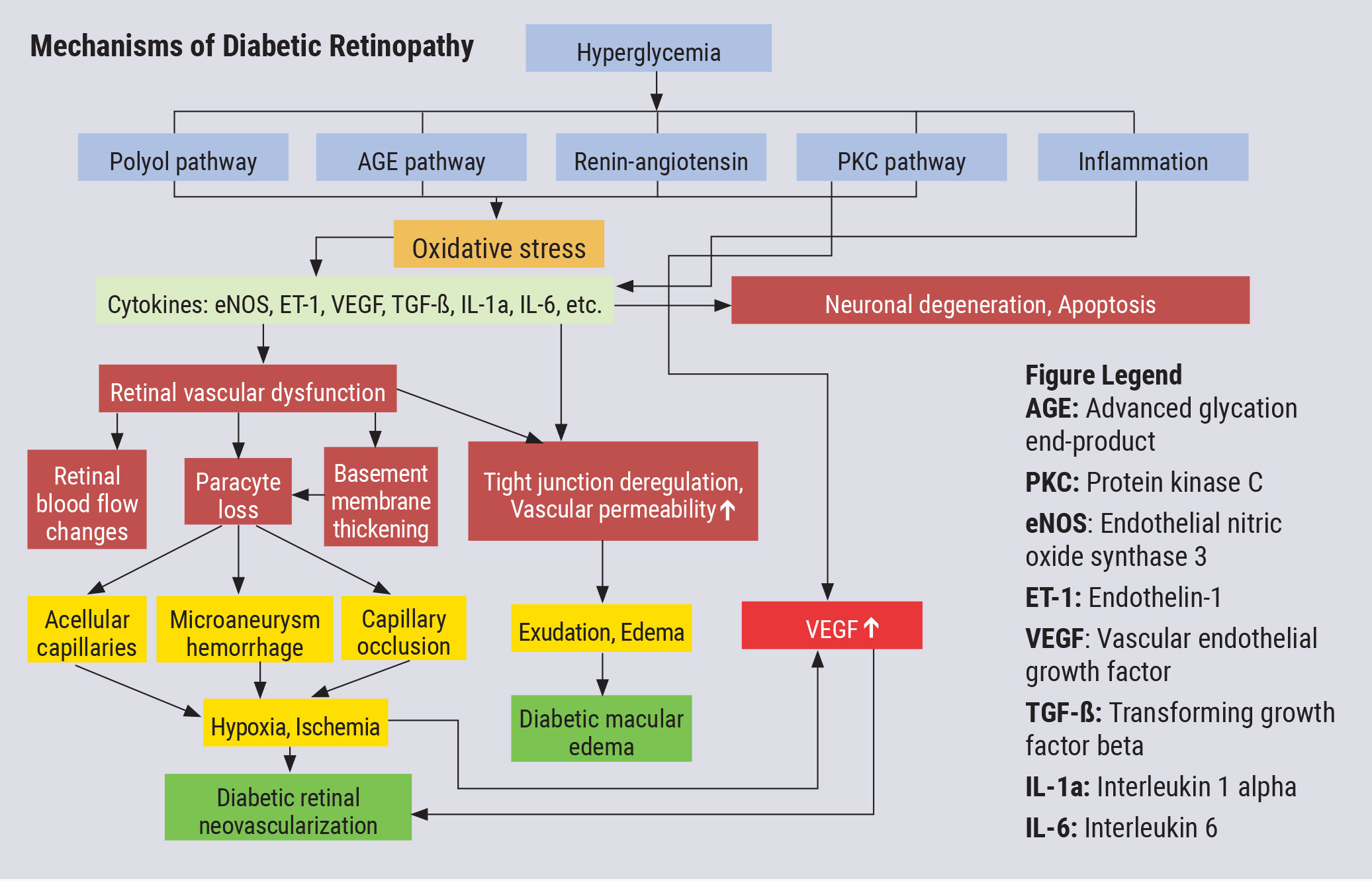 |
| This chart illustrates the complex pathophysiology of DR. Note that VEGF elevation happens fairly late in the cascade and directly before neovascularization, which partly explains why anti-VEGF therapy must be maintained indefinitely. Other interventions, especially those that might blunt activity further upstream from VEGF, could have a more long-lasting impact. Click image to enlarge. |
Risk Factors For DM/DR
Both DM and DR have the same risk factors, including family history of diabetes/DR, hypertension, cardiovascular disease, abnormal blood lipid level, prediabetes, gestational diabetes, obesity, longer DM duration, older age (>45), smoking, physical inactivity and race.
Nearly 100% of type 1 and 60% of type 2 diabetes patients will develop DR.6 Controlling for risk factors is one of the most important ways to reduce the incidence of diabetes complications. For example, the risk of DM microvascular complications is reduced by 40% for every 1% absolute decrease in A1c level.7,8 At the same time, the risk of diabetes-related microvascular complications is reduced by 12% for every 10mm Hg reduction in systolic blood pressure in hypertensive patients.9 Early detection of diabetes and appropriate treatment can reduce the risk of DR.
Pathophysiological DR Findings
The early physiological change in retinal vessels in DR—reduced retinal blood flow—starts from endothelial vasodilator dysfunction in retinal arterioles. The detailed mechanism is an impairment of the vasodilator, nitric oxide and overexpression of the vasoconstrictor. This decreased retinal blood flow leads to retinal tissue ischemia and, ultimately, to vessel leakage.10,11 The pathological changes in the retinal vessels in DR include basement membrane thickening, pericyte loss and epithelial tight junction loss. The last pathological change is the loss of endothelial cells coupled with capillary closure.
The pathological pathways of DR include both inflammatory and angiogenic processes, with both neuronal and vascular injury/dysfunction. The molecular pathways of DR include oxidative stress, with activation of the sorbitol pathway, renin-angiotensin system, diacylglycerol-protein kinase C pathway and advanced glycation endproduct pathway, as well as raised vascular endothelial growth factor (VEGF) levels.12 Chronic hyperglycemia and hypertension induce oxidative injury, then microthrombi formation followed by cell adhesion molecule activation/leukostasis and additional cytokine activation in a vicious cycle. All of these pathways/mechanisms induce further retinal damage.13 The detailed pathway can be found in the included table.
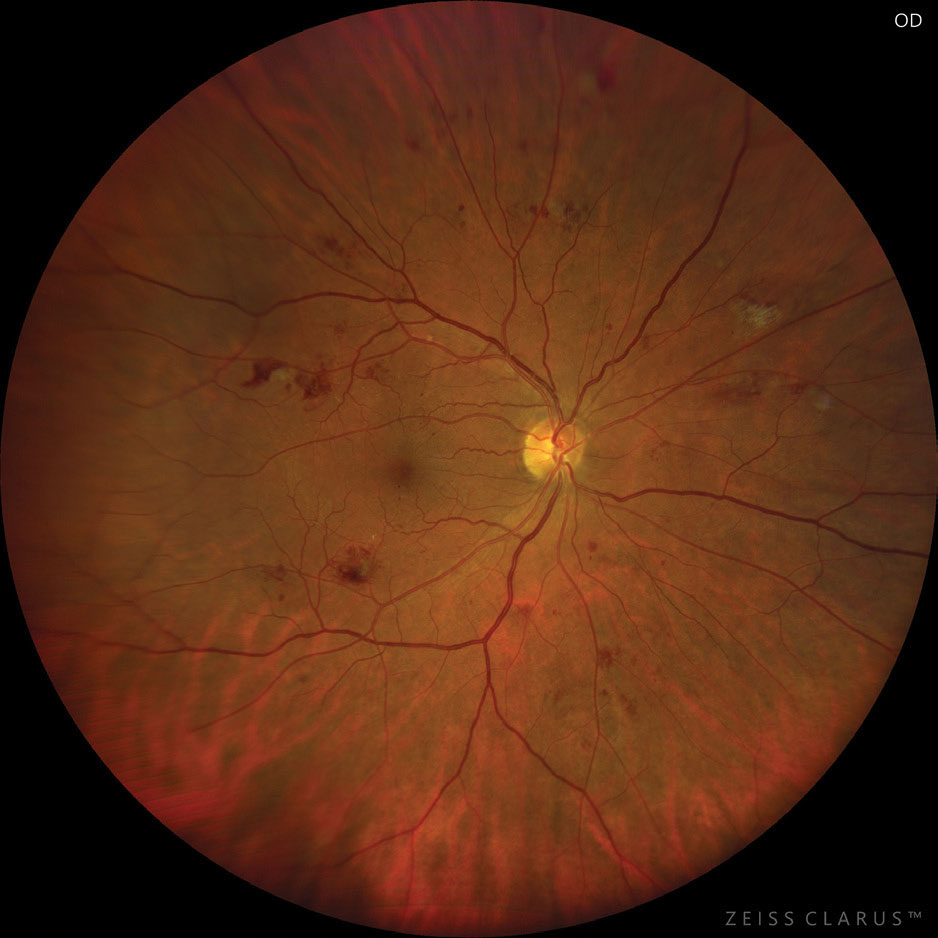 |
| Anti-VEGF may help improve visual outcomes for cases of moderate to severe NPDR. Photo: Jay M. Haynie, OD. Click image to enlarge. |
Prevalence of DR
In the United States, 40% of diabetes patients (age ≥40) have some degree of DR, whereas 8% have vision-threatening DR.14 The American Academy of Ophthalmology’s 2016 preferred practice patterns for DR state that about 60% of type 1 diabetes patients will develop any stage of DR within 10 years, whereas 53% to 84% of type 2 diabetes patients will develop DR within 19 years.15 Approximately 20% of type 1 diabetes patients and 14% to 25% of type 2 diabetes patients will develop diabetic macular edema (DME) after 10 years.16 The most common cause of vision loss in diabetes is DME.17
Stages of DR
To effectively care for their patients, optometrists need a clear understanding of the stages of DR. However, grading systems vary slightly depending on the source. For example, the International Council of Ophthalmology (ICO) published guidelines for diabetic eye care in 2017 and defined mild nonproliferative DR (NPDR) as “microaneurysms only.”18 Since the American Optometric Association (AOA) is one of the most prominent organizations supporting optometry, it is reasonable to follow the 2020 edition of the AOA clinical practice guidelines on DR staging, a modified version of the Early Treatment Diabetic Retinopathy Study (ETDRS).14,19
Mild NPDR. This stage includes at least one microaneurysm without additional significant retinopathy stages. Its underlying pathological features include increased vascular permeability due to the loss of capillary pericytes with vascular outpouching that results in its characteristic appearance. Microaneurysm formation is the earliest clinical characteristic of DR, but only 5% of mild NPDR cases will develop proliferative DR (PDR) within a year.
Moderate NPDR. This includes microaneurysms, dot or blot hemorrhages (from leaking microaneurysms or retinal capillaries in deeper retinal layers), cotton wool spots (formerly called “soft exudates,” arising from disruption of axoplasmic flow in the nerve fiber layer), mild venous beading (a biomarker of retinal ischemia) and intraretinal microvascular anomaly formation (IRMA). The latter phenomenon, IRMA, provides a “shunt” from the nonperfused retina to the perfused retina (either from remodeling of existing vessels or new growing vessels). IRMA is thought to be the “germ beds” of incipient neovascularization and commonly results in neovascularization elsewhere due to retinal ischemia. It always occurs within the intraretinal layers and is very difficult to detect in early stages without fluorescein angiography. Its role is to supply retinal areas of nonperfusion in DR.
Approximately 12% to 27% of moderate NPDR cases will develop PDR within one year. Vascular closure (retinal nonperfusion) typically develops in patients with greater than moderate NPDR.
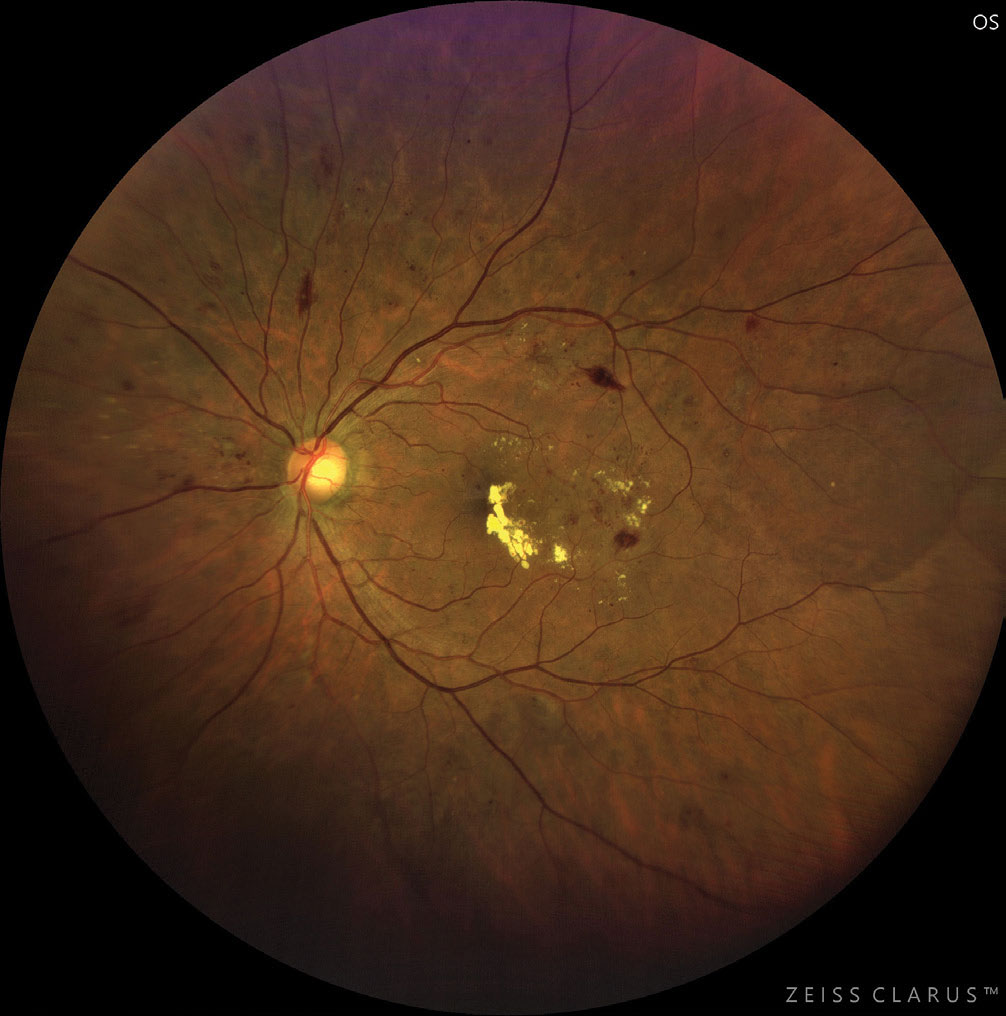 |
| This patient with moderate to severe NPDR developed CSME. Photo: Jay M. Haynie, OD. Click image to enlarge. |
Severe NPDR. This stage carries a high risk for development of PDR and consists of at least one of the following: severe microaneurysms and dot or blot hemorrhages in all four retinal quadrants, definite venous beading in two or more quadrants or prominent IRMA in one or more quadrants (4:2:1 rule). IRMA is very hard to be detected clinically; therefore, once it is visible the patient likely has prominent IRMA. Severe NPDR has a 50% risk of developing into PDR within one year.
Very severe NPDR. This stage includes two or more of the criteria of severe NPDR but without neovascularization. Very severe NPDR has a 75% of risk of developing into PDR in one year.
PDR. This stage is characterized by neovascularization (growth of new vessels) on the retinal surface, optic nerve or anterior segment (iris or angle). Neovascularization with inflammatory cells and fibrovascular proliferation may induce vitreous hemorrhage or retinal detachment.20 PDR staging in the 2020 AOA guidelines only consists of high-risk stages and doesn’t include mild and moderate stages, as does the 2017 ICO.19 High-risk PDR has at least three of the following four risk factors:
- Presence of new retinal vessels.
- Presence of new retinal vessels on or near the disc.
- Presence of any pre-retinal or vitreous hemorrhage.
- Presence of moderate or severe new retinal vessels or new vessels greater than half the disc area in size.
DME
This complication can occur during any stage of DR. It is characterized by intraretinal fluid leaking within the macula, with or without lipid exudate or cystoid changes, due to breakdown of the blood-retinal barrier. Common pathological changes associated with DME may include disruption of the perifoveal capillary network and capillary nonperfusion, widening of the foveal avascular zone and macular ischemia, though these features are also found in patients with DR without DME. The underlying mechanisms for DME include ischemia, oxygen-free radicals/oxidative stress that increase capillary permeability, production of pro-inflammatory cytokines and elevated levels of VEGF that further promote vascular leakage.
Clinically significant macular edema (CSME) is defined by at least one of the following three features:21
- Thickening of the retina at or within 500µm (or one-third of the disc diameter) of the center of the macula.
- Hard exudates at or within 500µm of the center of the macula associated with thickening of the adjacent retina.
- An area of retinal thickening one disc diameter or larger, part of which is within one disc diameter of the center of the macula.
It is important to note that the term “CSME” has largely become an anachronism with the advent of OCT. DME is now defined as center-involved (thickening or intra-retinal fluid within the central 1mm subfield on OCT) or non-center-involved (thickening or fluid within other OCT subfields).22
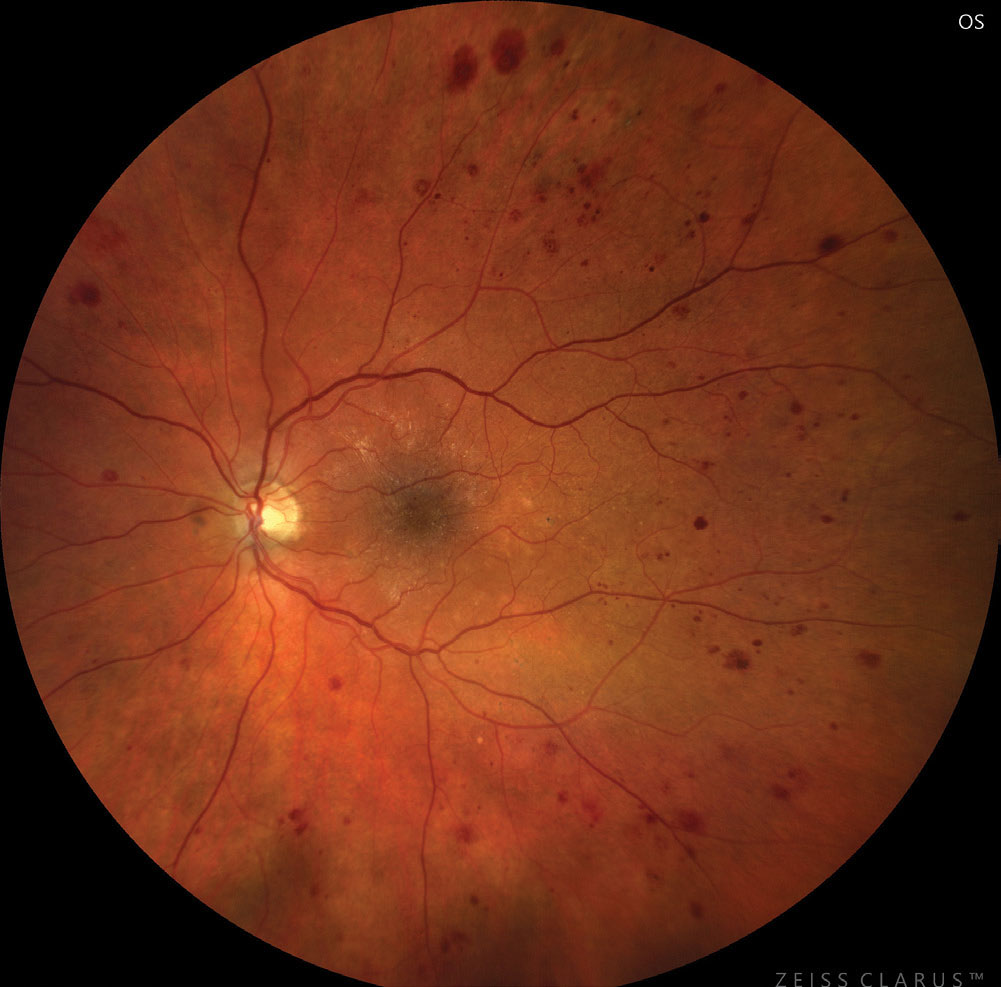 |
| Severe NPDR based on hemorrhages and microaneurysms in four retinal quadrants. Photo: Jay M. Haynie, OD. Click image to enlarge. |
Treatments For DR/DME
There are a number of medical and surgical treatment options available for these conditions, and it is important ODs recognize the best approach for each of their patients.
Intravitreal anti-VEGF injection. Ischemia induces VEGF production, which causes angiogenesis and induces retinal neovascularization in diabetes. In the endothelial cells of retinal vessels, VEGF regulates tight junctions, impairment of which damages the blood-retinal barrier. Given this biology, anti-VEGF injections were developed, tested and proved to help reduce retinal vascular leakage and neovascularization.23 The discovery of VEGF was a decade-long process.24
For DME, and depending on study-specific visual acuity gains or anatomic improvement of macular thickening, anti-VEGF injections demonstrate a 10% to 70% success rate, which is better than laser treatment.25,26 A general principle is that patients treated earlier for DME achieve superior outcomes. The use of anti-VEGF agents has been shown to preserve and improve vision in patients with DME.
The scope of treatment of VEGF in DR includes DME or CSME, PDR and severe or very severe NPDR, though both ranibizumab and aflibercept have FDA approval for treating any level of DR with or without DME. Faricimab-svoa was recently approved for the treatment of DME with dual action mechanisms to inhibit VEGF and angiopoiten.27
Anti-VEGF therapy is the initial treatment choice for center-involved macular edema with vision impairment, with possible subsequent or deferred focal laser treatment. Earlier treatment with these intravitreal injections could prevent vision-threatening complications from developing later on, reduce the need for potential future treatment and promote better visual acuity. Anti-VEGF injections can regress the severity of moderate to severe NPDR without DME and reduce the risk of developing DME and other sight-threatening complications related to retinal neovascularization, as shown in the PANORAMA study as well as DRCR.net Protocol W.28,29
Anti-VEGF treatment also showed noninferiority to panretinal photocoagulation (PRP) for visual acuity preservation in subjects with PDR in DRCR.net Protocol S, with secondary analysis showing far better preservation of the visual field (as expected) and significantly lower risk for developing center-involved DME or requiring vitrectomy in the anti-VEGF group.30 However, intravitreal injections of anti-VEGF agents still carry a risk for complications such as subconjunctival hemorrhage, vitreous hemorrhage, cataract, endophthalmitis, retinal tear/detachment, eye pain, secondary glaucoma, etc. If patient compliance is poor, there is an increased burden of care and need for concern.
Neovascularization of the iris involves the blood vessels growing on the anterior surface of the iris as a result of retinal ischemia. This condition is more likely to occur near the pupillary border. It is important to detect whether the angle is involved or not by performing gonioscopy. Hyphemas can be seen. Treatment for neovascularization of the iris is PRP with or without an anti-VEGF injection.
Neovascular glaucoma is a secondary form with neovascularization of the iris and high intraocular pressure (IOP). It is the result of posterior segment ischemia (diseases like PDR, central retinal vein occlusion, central retinal artery occlusion and ocular ischemic syndrome with abundant VEGF). Treating an ischemic retina includes anti-VEGF (induce the regression of neovascularization) and PRP.31 IOP treatments include IOP-lowering meds, glaucoma filtering surgeries, etc.
Key Principles to FollowExamination for DR. Perform dilated eye examination and communicate with the patient’s PCP on the ocular findings and surveillance recommendations. This is a vitally important job for every optometrist seeing patients with diabetes. Documentation. Make note of the severity of DR (if present) and whether DME or CSME is found. This not only facilitates recognition of disease progression but also reduces the risk of medico-legal liability. Educate patients. Compared with retinal specialists, optometrists can play a unique role in educating, monitoring and managing patients with diabetes. As primary eyecare providers, many diabetes patients see their OD annually and have developed close doctor-patient relationships that facilitate trust. Optometrists should spend some time educating patients about reducing the risk of diabetes and diabetic eye disease. ODs should also introduce possible treatment options. Patient education is time-consuming but will help promote compliance with annual eye examinations, as well as reduce the risk of both DR incidence and progression. Monitor patients. Follow-up with DM and DR patients based on recommendations from the AOA clinical practice guidelines and other evidence-based resources and in concert with the retinal specialist.37 Refer. Patients with severe NPDR, PDR or center-involved DME/CSME should be referred to a retinal specialist in a timely manner based on clinical practice guidelines for appropriate and possibly earlier treatment.14 Finally, refer patients with visual impairment to a vision rehabilitation specialist as appropriate when patients are experiencing difficulties with activities of daily living like self-monitoring of blood glucose levels, taking medications, reading and driving. |
Intravitreal corticosteroids. Steroids have been used in controlling both the inflammatory and angiogenic processes of diabetic retinal disease. Their use is generally limited to pseudophakic patients or those with persistent or refractory DME (typically in combination with anti-VEGF therapy). Sustained-release corticosteroids like dexamethasone or fluocinolone are commonly used in patients who exhibit a sub-optimal response to anti-VEGF, but they carry an increased risk of steroid-induced glaucoma.
Laser photocoagulation. Focal/grid macular laser treatment is limited to the treatment of focal, extrafoveal DME or for patients who show poor response to or cannot tolerate injections of anti-VEGF agents. PRP has been the standard of care for managing PDR and very severe NPDR by treating areas of capillary nonperfusion adjacent to areas of perfused retina, thereby lessening the production of VEGF, subsequent neovascularization and preventing retinal vessel leakage. PRP can be particularly valuable in conjunction with anti-VEGF therapy for patients with high-risk PDR.32
Vitrectomy. Surgical removal of the vitreous is used for treating vitreous hemorrhage, DR with fibrosis, tractional retinal detachments and vitreous traction with persistent DME. The complications of vitrectomy are similar to those of intravitreal injection of anti-VEGF agents. The risk of retinal detachment with anti-VEGF injections varied from tractional retinal detachment (5.2%) to rhegmatogenous retinal detachment (one per 7,500).33,34
Role of the OD
As primary eyecare providers, optometrists have an important role to play in every stage of the disease.
DM without retinopathy. ODs should be able to recognize the symptoms of DM, which can include thirst, hunger, fatigue, frequent urination, blurry vision, weight loss, slow wound healing and pain or numbness in the limbs, especially as some patients don’t know that they have the condition. The American Diabetes Association estimates that 7.5 million adults with type 2 diabetes are unaware they have diabetes. ODs should send these suspected DM patients to their PCP for evaluation. Even though many PCPs refer their patients with diabetes to optometrists for a dilated fundus exam, it is estimated, for example, that only 50% of Texas patients adhere to yearly eye exams.35 ODs should always encourage diabetes patients to get annual eye examinations and to do so on time.
ODs need to screen for a history of diabetes or prediabetes, perform a comprehensive dilated eye exam and educate patients about the risk factors for DR/DME. Additional education should include the possibility of refractive fluctuation with both poor and improving blood glucose control and the potential symptoms of retinal disease, though it is critical to teach patients that DR is asymptomatic at its earliest stages.
Finally, it is important that ODs send a diabetes eye exam report to the PCP and other diabetes care providers. For new diabetes patients, unbiased, evidence-based educational materials from the AOA, National Eye Institute, patient advocacy organizations and pharmaceutical companies can help build a good doctor-patient relationship.
Mild NPDR. Optometrists must communicate with PCPs and educate patients on the importance of controlling risk factors to reduce the risk of DR progression. Management includes glycemic control, smoking cessation, optimally managing both blood pressure and dyslipidemia, cardiovascular risk reduction, physical exercise and weight management. HbA1c accounts for 10% of DR risk, and hypertension plus dyslipidemia accounts for less than 10%.36,37 The probability of mild NPDR progression to PDR is 5% in one year per ETDRS data.14
The presence of hemorrhage anywhere, asymmetric hyperopic shift or any signs of DME warrants additional macular OCT/fundus photography. ODs should document whether the patient has DME/CSME or not. Referral to a retinal specialist within two to four weeks is indicated for center-involved DME and CSME. Patients should be followed up in one year or sooner if there is no DME, and sooner if their A1c is changing rapidly or they have poor metabolic control.14
Moderate NPDR. ODs should educate these patients about the importance of blood glucose/pressure/lipid control and the possibility of progression to PDR (12% to 27% of cases in one year). Fundus photography can detect most DR and document its severity and the presence of neovascularization. Macular OCT can facilitate the recognition of subtler DME. Communication with the PCP/internist/endocrinologist on ocular findings and recommendations/follow-up is critical. Follow-up every three to nine months in the absence of DME is warranted. Similar to mild cases, patients should be referred to a retinal specialist within two to four weeks if center-involved DME or CSME are present. Follow-up should be sooner if A1c is changing rapidly or in patients with poor metabolic control. Patients with non-center-involved DME should be watched closely—every one to six months—by the OD or referred to a retinal specialist.
All patients with DR or DME referred to a retinal specialist should also be reappointed with their optometrist to verify completion of the initial referral, ongoing therapy and surveillance for worsening disease and/or treatment-related adverse events (e.g., glaucoma, cataract, etc.). Evidence suggests that even retinal specialists tend to undergrade NPDR severity (accurate staging is best facilitated by widefield fluorescein angiography), so any patient with moderate or worse NPDR should be considered for referral as they may, in fact, have worse disease severity than that diagnosed by clinical examination.38
When to ReferUnderstanding how to best manage diabetes patients includes recognizing when a referral to a retinal specialist is necessary. The following patients need referral to a retinal specialist: center-involved DME or CSME, severe or very severe NPDR and PDR. The timeframe for referral for DME or CSME, irrespective of DR severity, is within two to four weeks. This is also applicable to patients with severe or very severe NPDR or non-high-risk PDR. Neovascularization of the iris and neovascular glaucoma need to be referred ASAP. The timeframe reduces to 24 to 48 hours for high-risk PDR.14 |
Severe NPDR/very severe NPDR/non-high-risk PDR. As these stages of DR portend much higher risk of severe vision loss, patients require careful education and appropriate referral. In addition to routine dilated fundus exam, macular OCT and fundus photography to document disease severity, gonioscopy should be performed if there is any sign of retinal or iris neovascularization to rule out angle neovascularization and help diagnose secondary glaucoma (always measure IOP in these patients). Document the stage of DR and presence of any DME/CSME (more likely as DR severity worsens) carefully. Communicate with the PCP/internist/endocrinologist/cardiologist/nephrologist about the ocular finding, referral to a retina specialist and probable treatment.
The PANORAMA study showed that proactive intravitreal aflibercept for moderately severe to severe NPDR cases reduces the risk of progression to vision-threatening complications. It also showed that good blood glucose control was not protective against subsequent progression to PDR, center-involved DME or anterior segment neovascularization in patients with moderately severe or severe NPDR, so don’t base referral decisions on diabetes control, but rather, disease severity. Patients with multiple complications, especially cardiovascular disease, poor cognition, multiple comorbidities and shortened lifespan, should not be instructed to attempt tight blood glucose control without the oversight of their treating diabetes physicians. These patients should be referred to a retinal specialist within two to four weeks, per AOA clinical practice guidelines.
While it remains a clinical judgement call for physicians on the most appropriate time to refer severe-stage patients without macular edema, the shift continues to move toward earlier referral and earlier intervention with anti-VEGF due to findings of PANORAMA and Protocol W. The AOA clinical practice guidelines currently state “follow-up every three to four months in consultation with an ophthalmologist experienced in the management of diabetic retinal disease is advisable for patients with severe or very severe NPDR.” Follow-up with the OD is prudent to ensure completion of the initial referral, ongoing therapy and surveillance for worsening disease and/or treatment-related adverse events.
High-risk PDR. These patients require prompt referral to a retinal specialist within 24 to 48 hours for treatment to preserve vision and reduce the high probability of blindness over five years (50%). Optometrists should continue to follow these patients to ensure adherence to the treatment regimen, address any primary care needs and provide or refer for vision rehabilitation, if necessary.
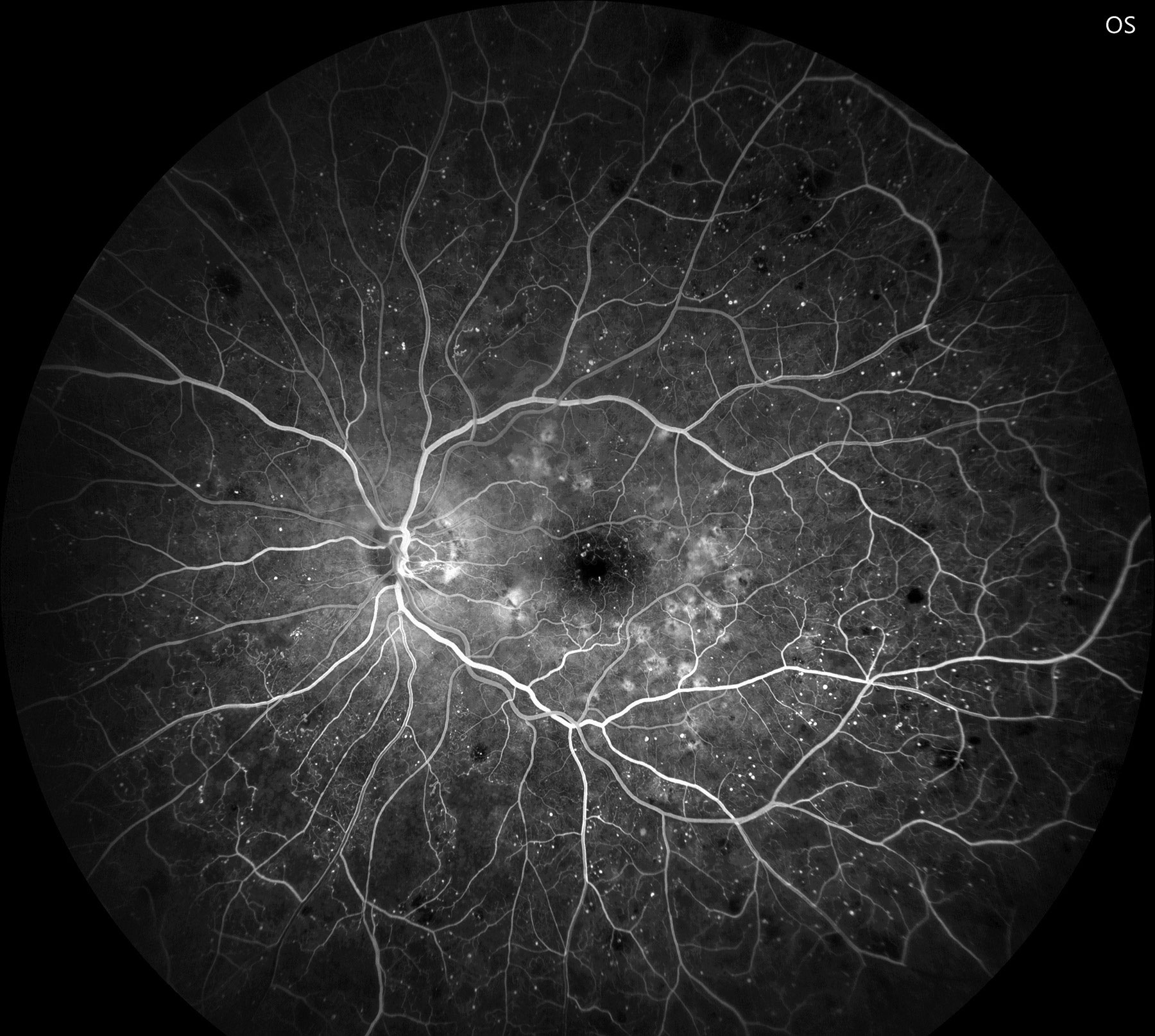 |
| Fluorescein angiography confirms early PDR with significant non-perfusion and neovascularization of the disc. Photo: Jay M. Haynie, OD. Click image to enlarge. |
Telescreening and Artificial Intelligence (AI)
These technologies will likely assist with earlier identification and accurate staging of diabetic retinal disease. Two AI systems are currently approved in the United States for autonomous detection and grading of DR and DME, and multiple other systems are in development with varying degrees of sensitivity and specificity.
Teleretinal DR screening programs are another option for reaching underserved populations. They have been employed for more than a decade and require a photographer/technician to capture retinal images (often without dilation) which are then sent to a certified image reading center where ophthalmologists, optometrists and clinical personnel judge the presence or absence of DR/DME, grade DR severity and personalize decisions for follow-up, in-person clinical examination and referral to a retinal specialist.
Teleretinal screening may be faster and more accessible, often reducing travel time, and can be more convenient than traditional in-office examination. However, it also has shortcomings, including equipment and training expenses and non-readable or non-gradeable images due to small pupil size, media opacities or poor field of view.
Takeaways
ODs are in the perfect position to play a key role in the management of diabetes patients. Therefore, they must have a comprehensive understanding of not only the disease but also what steps to take during every stage.
Dr. Yuen practices as a senior OD at the Central Texas VA Health Care System in Temple, TX. He holds a PhD degree and is a fellow of the American Academy of Optometry. He has no relevant financial interests to disclose.
The author thanks Paul Chous, OD, for his contributions to this article.
1. National Diabetes Statistics Report. https://www.cdc.gov/diabetes/data/statistics-report/index.htmlCDC. Accessed March 6, 2022. 2. National Diabetes Statistics Report 2020. Estimates of Diabetes and Its Burden in the United States. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed March 6, 2022. 3. What is Diabetes? https://www.cdc.gov/diabetes/basics/diabetes.html. Accessed March 6, 2022. 4. Galicia-Garcia U, Benito-Vicente A, Jebari S, et al. Pathophysiology of Type 2 Diabetes Mellitus. Int J Mol Sci. 2020;30;21(17):6275. 5. Ribhi Hazin, Mohammed K Barazi, Michael Summerfield. Challenges to establishing nationwide diabetic retinopathy screening programs. Curr Opin Ophthalmol. 2011;22(3):174-9. 6. Fong DS, Aiello L, Gardner TW, et al. Diabetic Retinopathy. Diabetes Care. 2003;26(suppl_1):s99–s102. 7. American Diabetes Association. Implications of the Diabetes Control and Complications Trial. Diabetes Care. 2003;26(Suppl 1):S25-7. 8. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet. 1998;12;352(9131):837-53. 9. UK Prospective Diabetes Study Group. Efficacy of atenolol and captopril in reducing risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 39. Br Med J. 1998;317:713-20. 10. Haefliger IO, Flammer J, Beny JL, et al. Endothelium-dependent vasoactive modulation in the ophthalmic circulation. Prog Retin Eye Res. 2001;20:209-25. 11. Hein TW, Potts LB, Xu W, et al. Temporal development of retinal arteriolar endothelial dysfunction in porcine type 1 diabetes. Invest Ophthalmol Vis Sci. 2012;53(13):7943-9. 12. Lechner J, O’Leary OE, Stitt AW. The pathology associated with diabetic retinopathy. Vision Res. 2017;139:7-14. 13. Grant MB, Afzal A, Spoerri P, et al. The role of growth factors in the pathogenesis of diabetic retinopathy. Expert Opin Investig Drugs. 2004;13(10):1275-93. 14. AOA. Evidence-based Clinical Practice Guideline: Eye Care of the Patient with Diabetes Mellitus. aoa.uberflip.com/i/1183026-evidence-based-clinical-practice-guideline-eye-care-of-the-patient-with-diabetes-mellitus-second-edition/0?m4=. October 4, 2019. Accessed March 6, 2022. 15. Diabetic Retinopathy PPP — Updated 2016. American Academy of Ophthalmology website. www.aao.org/preferred-practice-pattern/diabetic-retinopathy-ppp-updated-2016. Accessed March 6, 2022. 16. Klein R, Knudtson MD, Lee KE, et al. The Wisconsin Epidemiologic Study of Diabetic Retinopathy XXIII: the twenty-five-year incidence of macular edema in persons with type 1 diabetes. Ophthalmology. 2009;116(3):497-503. 17. Mitchell P, Annemans L, Gallagher M, et al. Cost-effectiveness of ranibizumab in treatment of diabetic macular oedema (DME) causing visual impairment: evidence from the RESTORE trial. Br J Ophthalmol. 2012;96(5):688-93. 18. International Council of Ophthalmology: Updated 2017 ICO guidelines for diabetic eye care. https://www.idf.org/component/attachments/attachments.html?id=407&task=download. Accessed May 16, 2022. 19. Treatment Diabetic Retinopathy Study Research Group. Grading diabetic retinopathy from stereoscopic color fundus photographs--an extension of the modified Airlie House classification. ETDRS Report Number 10. Ophthalmology. 1991;98(5 Suppl):786-806. 20. Klaassen I, van Geest RJ, Kuiper EJ, et al. The role of CTGF in diabetic retinopathy. Exp Eye Res. 2015;133:37-48. 21. Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch Ophthalmol. 1985;103(12):1796-806. 22. Waheed NK, Duker JS. OCT in the Management of Diabetic Macular Edema. Curr Ophthalmol Rep. 2013; https://doi.org/10.1007/s40135-013-0019-z. 23. Gupta N, Mansoor S, Sharma A, et al. Diabetic retinopathy and VEGF. Open Ophthalmol J. 2013;7:4-10. 24. Ferrara N. VEGF and Intraocular Neovascularization: From Discovery to Therapy. Transl Vis Sci Technol. 2016;5(2):10. 25. Nguyen QD, Brown DM, Marcus DM, et al. Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 2012;119(4):789-801. 26. Fong AH, Lai TY. Long-term effectiveness of ranibizumab for age-related macular degeneration and diabetic macular edema. Clin Interv Aging. 2013;8:467-83. 27. Vabysmo - FDA prescribing information, side effects and uses (drugs.com). https://www.drugs.com/pro/vabysmo.html. Accessed May 16, 2022. 28. ClinicalTrials.gov. Study of the efficacy and safety of intravitreal (IVT) aflibercept for the improvement of moderately severe to severe nonproliferative diabetic retinopathy (NPDR) (PANORAMA). March 24, 2016. clinicaltrials.gov/ct2/show/results/NCT02718326?cond=diabetic+Aflibercept&draw=2&rank=18. Accessed March 6, 2022. 29. Anti-VEGF Treatment for Prevention of PDR/DME. https://clinicaltrials.gov/ct2/show/NCT02634333?term=DRCR. Accessed May 16, 2022. 30. Gross JG, Glassman AR, Liu D, et al. Five-year outcomes of panretinal photocoagulation vs. intravitreous ranibizumab for proliferative diabetic retinopathy: a randomized clinical trial. JAMA Ophthalmol. 2018;136(10):1138-48. 31. Eid TM, Radwan A, el-Manawy W, et al. Intravitreal bevacizumab and aqueous shunting surgery for neovascular glaucoma: safety and efficacy. Can J Ophthalmol. 2009;44:451-6. 32. Gao S, Lin Z, Shen X. Anti-Vascular Endothelial Growth Factor Therapy as an Alternative or Adjunct to Pan-Retinal Photocoagulation in Treating Proliferative Diabetic Retinopathy: Meta-Analysis of Randomized Trials. Frontiers in Pharmacology. 2020; doi:10.3389/fphar.2020.00849. 33. Arevalo JF, Maia M, Flynn HW Jr, et al. Tractional retinal detachment following intravitreal bevacizumab (Avastin) in patients with severe proliferative diabetic retinopathy. Br J Ophthalmol. 2008;92(2):213-6. 34. Storey PP, Pancholy M, Wibbelsman TD, et al. Rhegmatogenous Retinal Detachment after Intravitreal Injection of Anti-Vascular Endothelial Growth Factor. Ophthalmology. 2019;126(10):1424-31. 35. The Quest for Quality: It Takes a Team to Treat a Patient, TexasMedicine Cover Story October 2000. https://app.texmed.org/tma.archive.search/961.html. Accessed May 16, 2022. 36. Hirsch IB, Brownlee M. Beyond hemoglobin A1c--need for additional markers of risk for diabetic microvascular complications. JAMA. 2010;303(22):2291-2. 37. Klein R, Knudtson MD, Lee KE, et al. The Wisconsin Epidemiologic Study of Diabetic Retinopathy: XXII the twenty-five-year progression of retinopathy in persons with type 1 diabetes. Ophthalmology. 2008;115(11):1859-68. 38. O’Connor PM, Harper CA, Brunton CL, et al. Shared care for chronic eye diseases: perspectives of ophthalmologists, optometrists and patients. Med J Aust. 2012;196(10):646-50. |
