Pregnancy And the Eye: What To Do When Caring for Two
Clinicians must understand how to manage ocular changes and treatment contraindications.
By Cecelia Koetting, OD
 |
Release Date: June 15, 2020
Expiration Date: June 15, 2023
Estimated Time to Complete Activity: 2 hours
Jointly provided by Postgraduate Institute for Medicine (PIM) and Review Education Group
Educational Objectives: After completing this activity, the participant should be better able to:
- Understand the ocular changes pregnancy can induce.
- Manage the ocular changes associated with pregnancy.
- Determine when to treat an ocular condition and when to wait.
- Successfully comanage with a patient’s obstetrician.
Target Audience: This activity is intended for optometrists engaged in the care of patients who are pregnant or nursing.
Accreditation Statement: In support of improving patient care, this activity has been planned and implemented by the Postgraduate Institute for Medicine and Review Education Group. Postgraduate Institute for Medicine is jointly accredited by the Accreditation Council for Continuing Medical Education, the Accreditation Council for Pharmacy Education, and the American Nurses Credentialing Center, to provide continuing education for the healthcare team. Postgraduate Institute for Medicine is accredited by COPE to provide continuing education to optometrists.
Faculty/Editorial Board: Cecelia Koetting, OD, Virginia Eye Consultants, Norfolk, VA.
Credit Statement: This course is COPE approved for 2 hours of CE credit. Course ID is 68213-GO. Check with your local state licensing board to see if this counts toward your CE requirement for relicensure.
Disclosure Statements:
Dr. Koetting has no disclosures.
Managers and Editorial Staff: The PIM planners and managers have nothing to disclose. The Review Education Group planners, managers and editorial staff have nothing to disclose.
When pregnant patients present for eye care, clinicians must be up to date on the everchanging do’s and don’ts of clinical care. Just as clinicians comanage patients with other specialties, they should also feel comfortable reaching out to patients’ obstetricians.
When seeing any female of child-bearing age, clinicians should ask if they are pregnant, nursing or thinking of becoming pregnant.
Medications
Historically, drugs were labeled based on a category, with category A being the safest medications to take during pregnancy.1,2 Category B included those showing no risk in animal studies, but a lack of controlled studies on pregnant women.1,2 Category C indicated that animal studies showed risk to the fetus, but no human studies have been performed or are not available.1,2 Category D indicated positive evidence of potential fetal risk but benefits for the pregnant woman may be acceptable despite the risk.1,2 Lastly, category X was contraindicated in women who are pregnant or may become pregnant because of known fetal risk and abnormalities.1,2
In 2015, the FDA implemented new pregnancy and lactation guidelines to help healthcare providers better assess benefit vs. risk. The updated labeling system removes the letter categories and instead provides narrated sections on risk for women who are pregnant and breastfeeding, as well as men and women of reproductive age.3 Many clinicians still think of drugs based on their historic category, and this article includes categories when available.
When prescribing, keep in mind which medications can and cannot be administered.
Systemic medications. Among the oral antibiotics, which may be needed for gland and soft tissue infections, it is safe to use Augmentin (amoxicillin and clavulanate, GlaxoSmithKline), erythromycin, azithromycin and amoxicillin, which are all historic category B.1,4 Commonly used oral antibiotics such as doxycycline, tetracycline, sulfonamides, trimethoprim, neomycin and fluoroquinolones are historic category C or D and should be avoided.1,4
For patients with herpes simplex virus or shingles, oral antivirals acyclovir, valacyclovir and famciclovir are all historic category B.1,4 However, acyclovir is the only one approved for use in lactating women.1,4
When oral steroids are needed, clinicians should consult with the patient’s obstetrician because this class of medication is considered historic category C.5 Fetal changes such as cleft palate, heart defects and neural tube defects have been noted with the use of systemic corticosteroids.5
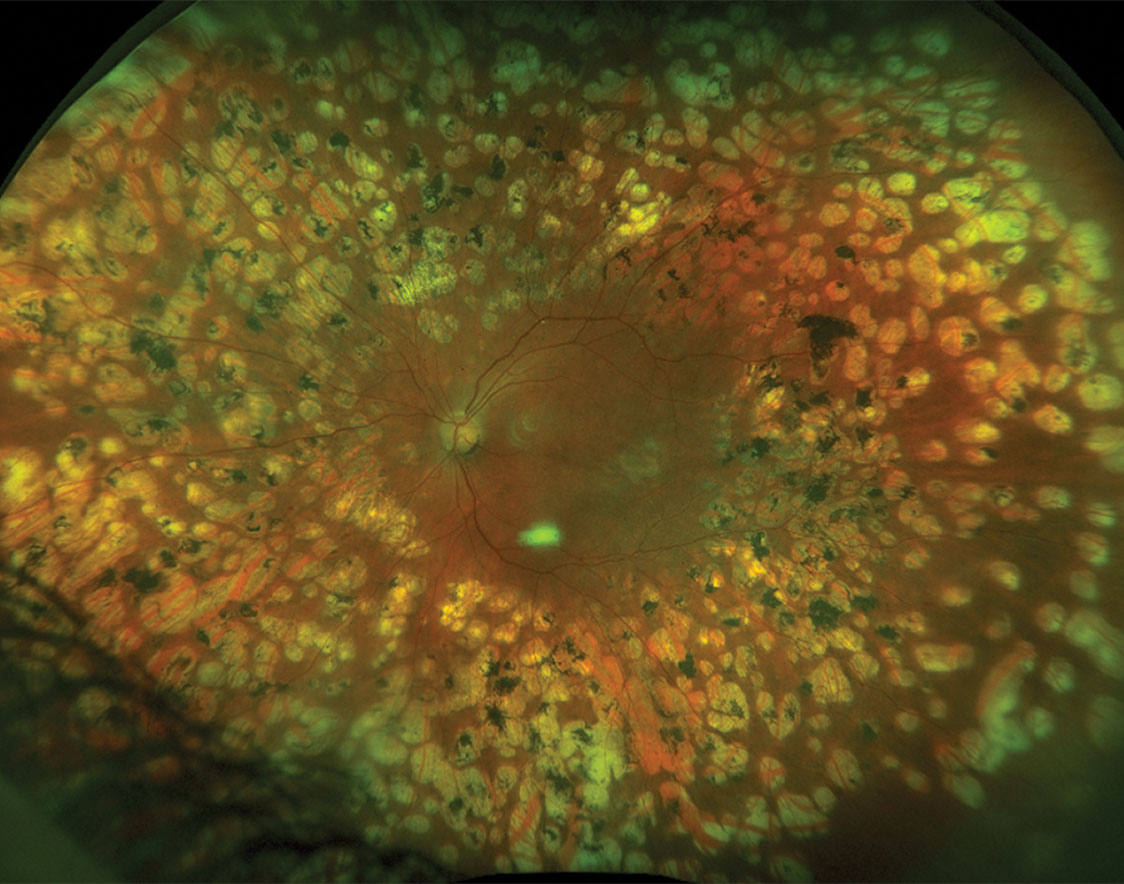 |
| Fig. 1. This pregnant patient has had significant panretinal photocoagulation treatment for proliferative diabetic retinopathy, leaving scaring 360° throughout the peripheral retina. Because of her history of poorly controlled Type 1 diabetes, she was monitored every three months with dilation for re-activation or progression. Click image to enlarge. |
When a patient is in need of pain medication, acetaminophen is historic category B in all three trimesters and is considered safe.6 Other over-the-counter (OTC) medications, such as aspirin and ibuprofen, are historic category D in at least one of the trimesters and should be avoided.6 In patients who are breastfeeding acetaminophen and ibuprofen are safe to use but aspirin and naproxen are typically avoided.6 Hydrocodone (historic category C) and oxycodone (historic category B) are sometimes given to women to help with pain after birth.7 Although they are the preferred pain medications when OTC medications aren’t enough, both have been found in breast milk and can affect the baby.6 If considering recommending these, consult with the patient’s obstetrician.
Topical medications. Similar to systemic medication, fluoroquinolones are typically avoided unless the benefit outweighs the risk, such as in the case of resistant bacteria with fluoroquinolone sensitivity. If this prescription is necessary, clincians must consult the patient’s obstetrician.
Tobramycin is historic category B, as is erythromycin, polymyxin B and topical azithromycin. In the case of severe ulcers or bacterial keratitis, fortified cephalosporins are also historic category B and are safe to use in this patient population.
Corticosteroids are often used to control a patient’s inflammation, whether it be anterior surface or intraocular. All ocular formulations of corticosteroids are historic category C. Optometrists are familiar with the risks to their patients such as steroid-induced cataracts and intraocular pressure (IOP) increase.5 No published studies have associated fetal changes, such as cleft palate, heart defect and neural tube defect, with topical ophthalmic corticosteroids.5 Regardless, clinicians should discuss their use with the patient’s obstetrician before initiating or continuing any steroid therapy.
For allergies, the only historic category B ocular allergy medication is Lastacaft (alcaftadine, Allergan). Anti-inflammatory medications for dry eye, such as cyclosporine A, are historic category C and come with concerns of teratogenic and fetal abnormalities; thus, the patient’s obstetrician should be consulted first. Lifitegrast has no human data available; however, an animal study shows fetal changes with intravenous (IV) administration.8
Most glaucoma medications are historic category C. Prostaglandins are a cause for concern, as research shows they can induce labor and miscarriage.5 Beta blockers are cautioned in the first trimester as well as shortly before birth to avoid neonatal beta blockade.5 Carbonic anhydrase inhibitors have teratogenic and hepatorenal effects.5
The rho-kinase inhibitor Rhopressa (netarsudil, Aerie Pharmaceuticals) currently has no clinical data regarding use in pregnant women or its presence in breast milk, although animal data shows some teratogenic effects.9 Oral Diamox (acetazolamide, Duramed Pharmaceuticals) is also historic category C and can cause fetal abnormalities.5 Alphagan (brimonidine, Allergan), unlike the others, is historic category B and safe to use in all trimesters. However, it should be discontinued prior to breastfeeding due to concerns of sleep apnea and central nervous system (CNS) depression in infants.
Because IOP decreases during pregnancy, patients who are on one ocular medication may be stable without it, but should be monitored closely. In cases where an ocular medication is needed, brimonidine is a safe option, but the patient must stop while breastfeeding.
Other options such as minimally invasive glaucoma surgery or selective laser trabeculoplasty may be considered in some patients.
Although not widely necessitated by women of child-bearing age, intravitreal anti-vascular endothelial growth factor (VEGF) treatment is needed for some retinal diseases such as proliferative diabetic retinopathy, neovascular glaucoma or chorioretinal neovascularization. During pregnancy, VEGF plays an important role for fetal and placental vasculature, and reduction has been linked with defective embryogenesis and fetal loss in humans.10
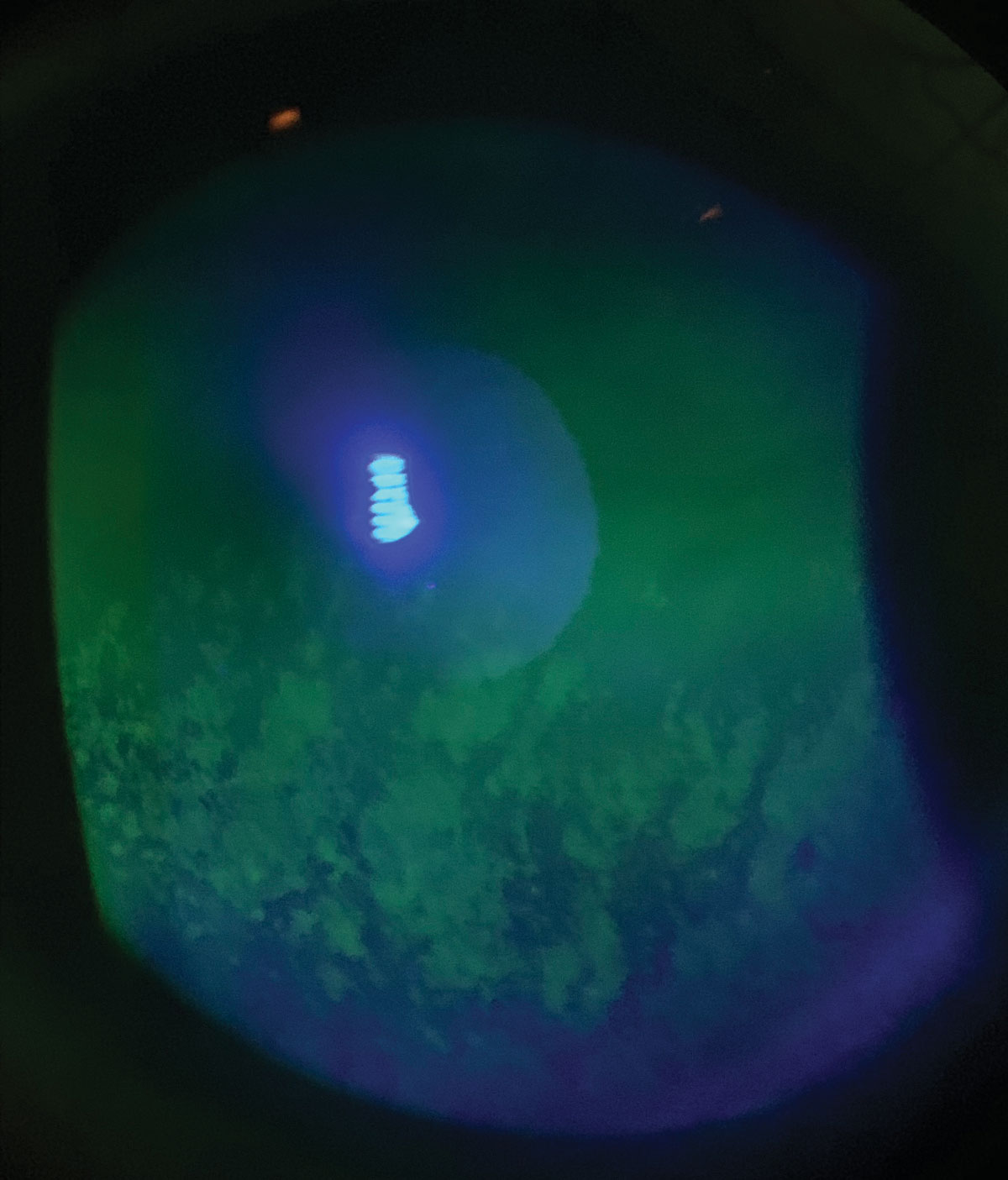 |
| Fig. 2. This patient experiened exposure keratopathy secondary to Bell’s palsy shortly after giving birth. She has a defined area of punctate keratopathy staining on the lower third of her cornea. Click image to enlarge. |
The use of anti-VEGF drugs during pregnancy may potentially cause systemic side effects in the mother and harm to the fetus, including spontaneous miscarriage and preeclampsia.10 Therefore, anti-VEGF should only be used in cases where the potential benefit to the patient justifies the potential risk to the fetus. This requires consultation with the patient’s obstetrician and careful patient education on the possible effects of the drug.
Breast milk contains VEGF to help with the infant’s development and maturation of their digestive system.11 Although the labeling of the anti-VEGF agents ranibizumab and aflibercept indicate that it is unknown if the drugs are excreted in breast milk, a recent study found that they are detected after intravitreal injection.11 This raises safety concerns for the patient’s infant. Women who are breastfeeding should be counseled prior to receiving treatment, including a discussion regarding risk vs. benefit and possible cessation of breastfeeding.
Dilation: Yes or No?
In general, occasional dilation is acceptable, but repeated dilation is avoided in pregnant and breastfeeding patients because of risks to the fetus and newborn.4 There are times when the benefits outweigh the risks and it is appropriate and necessary to dilate. Optometrists should not hesitate to have a discussion with a patient’s obstetrician prior to administering medication.
When dilating, consider using shorter acting agents such as tropicamide 0.5% (historic category C) vs. 1%. Clinicians can have the patient punctal occlude after drops are administered or put in temporary punctal plugs to decrease systemic absorption. Avoidance of phenylephrine is suggested since systemic use of the drug may cause minor malformations during first trimester use as well as fetal hypoxia and bradycardia.12
If a patient is breastfeeding they should be advised that it is unknown if cycloplegics and mydriatics are excreted in human breastmilk.12 However, low-weight infants are susceptible to systemic hypertension when 10% or 2.5% phenylephrine eye drops are used, so they should be avoided.13 Atropine and homatropine may also cause minor fetal malformations when they are used systemically.4,12
Fluorescein and indocyanine green dyes both cross the placenta, although no reports of teratogenic effects on animals exist and no adverse effects are reported in humans.12,14 Most vitreoretinal specialists avoid these tests on pregnant patients unless absolutely necessary.14
Specialists suggest that, despite the low toxicity, the mother avoid nursing for eight to 12 hours after topical fluorescein use since it has been detected in breast milk.12,15
Ocular Changes and Complications
Pregnancy can induce a number of ocular changes that optometrists must understand to ensure they can manage patient’s successfully.
Refractive changes. During pregnancy, a patient’s prescription and vision may change. More commonly it is a myopic shift, but hyperopic shifts may also occur. Studies show that approximately 40% to 75% of pregnant women experience a change in their distance vision.16-19 Researchers found that during the second trimester 51% of patient’s distance vision was affected and 11% of patient’s near vision was affected.16 By the third trimester the same study showed 74.7% of patient’s distance vision was affected and 20.2% of patient’s near vision.16 Postnatal, only 8.2% of patients still had a change in distance vision and 4% in near vision.16
Most patients’ refractive changes do resolve and are not long-term. Other studies had similar outcomes noting that most women’s vision returned to pre-pregnancy refractive error shortly after birth and cessation of breastfeeding.17
These refractive changes are thought to be related to hormonal changes resulting in fluid retention in the cornea; however, causality requires further study.16
Less often, refractive shifts can occur because of cataract changes, diabetes, preeclampsia or an accommodative spasm. Both insufficiency and paralysis of accommodation during lactation are possible.18,19 These typically resolve shortly after breastfeeding is discontinued, but may require vision therapy.
Dry eye disease. During pregnancy, this condition can worsen or develop.4 One cause of this is likely physical dehydration due to nausea and vomiting, especially during the first trimester.1 In addition, changes in the cornea and lacrimal system can lead to changes in tear film physiology.20 There is a possible immune reaction to the lacrimal duct cells leading to destruction of acinar cells by prolactin.20
With the worsening of dry eye disease, secondary contact lens intolerance and discomfort are likely. This may be exacerbated by decreased corneal sensitivity common throughout pregnancy, peaking in the third trimester.18,19 Sensitivity returns to normal eight weeks postpartum.18,19
Treatment can include adding punctal plugs and artificial tears, omega-3 and refitting soft contact lenses. Remember that topical ocular cyclosporine and steroids are historic category C, so these medications should be used only when absolutely necessary. However, it is important to consult with the patient’s obstetrician prior to prescribing.
Adnexal changes. Chloasma, also known as pregnancy mask, is increased pigmentation around the eyes and cheeks. This condition, caused by increased estrogen, progesterone and melanocyte stimulating hormones, will fade over time postpartum.18,19 Another occasional adnexal change during or after pregnancy is unilateral ptosis. This is, in theory, caused by defects in the levator aponeurosis from fluid, hormones and other changes caused by the stress of labor and delivery.18,19
Graves’ disease. This condition is the most common cause of hyperthyroidism during pregnancy, leading to unilateral and bilateral proptosis.18,19 Patients with preexisting Graves’ disease may note an exacerbation during the first trimester that subsides during the rest of the pregnancy, only to flare up again postpartum.18,19 Treatment is similar to that used for non-pregnant women, and ocular sequelae should be managed to decrease discomfort.18,19
Preeclampsia and eclampsia. Preeclampsia is classified by a triad of symptoms in a normotensive pregnant woman: BP >140/90mm Hg, edema and proteinuria after 20 weeks of pregnancy. When the patient has the triad along with contractions, it is considered eclampsia.
Of the 5% of pregnant women who develop these conditions, one in three have ocular sequelae such as blurred vision, photopsia, scotoma and diplopia.18 Up to 60% of those with preeclampsia or eclampsia will have hypertensive retinopathy with retinal arteriolar narrowing.19
If these changes are noted and the patient is undiagnosed, clinicians must convey these findings to the patient’s obstetrician and monitor them during and after pregnancy. Severity of ocular symptoms is directly related to the severity of the preeclampsia.19 Typically these changes dissipate postpartum.
In less than 1% of preeclamptic patients and 10% of eclamptic patients, exudative retinal detachment may occur.19,21,22 The cause is thought to be choroidal ischemia, showing a delayed filing of choriocapillaris in the presence of normal retinal vasculature on intravenous fluorescein angiography.19,22
Approximately 10% of severe preeclampsia patients develop hemolysis, elevated liver enzymes and low platelets (HELLP).19,22 These patients will develop bilateral serous retinal detachments with yellow/white subretinal opacities and possible vitreous hemorrhage.19,22 Unfortunately, this syndrome is associated with poor prognosis for both mother and fetus. When a patient presents with these findings, clinicians must discuss them with the patient’s obstetrician. They should also be referred to a retinal specialist for possible treatment of the ocular sequelae.
Preeclamptic patients with choroidal infarcts may also develop Elschnig spots that will resolve after delivery.19,21,23 Infrequently, cortical blindness lasting four to eight hours associated with preeclampsia/eclampsia can occur as a result of petechial hemorrhages and focal edema in the occipital cortex.24
Diabetes. Patients who develop diabetes during pregnancy—gestational diabetes—typically return to normal after delivery.25 This usually occurs during the second to third trimester and glucose tolerance will return to normal approximately six weeks after giving birth (Figure 1).
These patients are at a higher risk of developing Type 2 diabetes during their lifetime and should be monitored with yearly dilated eye exams.25 During pregnancy, given the relative shortness of the disease, these patients are at a small risk of developing diabetic retinopathy (DR).18.19 According to the American Optometric Association (AOA) practice guidelines, retinal evaluation is not indicated for these patients.26
Patients who had Type 1 or Type 2 diabetes prior to pregnancy have a much higher risk of developing complications related to their diabetes during pregnancy. Patients who already have DR prior to pregnancy will note a quicker progression.27
According to one study, those with moderate to severe non-proliferative DR (NPDR) show a 54.8% progression during pregnancy.27 The same study shows only a 21.1% progression in disease process for those with mild NPDR.27 Approximately 22% of those with severe NPDR will progress to proliferative DR (PDR).18 Up to 45% of those who already have PDR will note worsening of the disease. The treatments are the same as those for non-pregnant patients.
To monitor for these changes, any diabetic patient who is pregnant or thinking of becoming pregnant should have regular eye exams and be counseled about their increased risk of DR progression both during and after pregnancy. Patients with Type 1 and Type 2 diabetes should have a comprehensive eye and vision exam prior to a planned pregnancy and once during every trimester with follow-up at six to 12 months postpartum, according to the AOA practice guidelines.26
Bell’s palsy. While pregnancy isn’t a cause of Bell’s palsy, it does lead to a higher risk of occurrence, especially during the third trimester and within the first weeks after giving birth.28 This condition, caused by compression or inflammation and swelling of the facial nerve, has rapid onset of weakness or total paralysis on one side of the face. Typically, the face will droop and the patient will experience changes in the amount of tears and saliva produced. People generally recover with or without treatment, but both oral corticosteroids and antiviral drugs may be used. Resolution will occur over a few weeks to six months.29
With the paralysis leading to poor lid closure and incomplete blink, clinicians should be concerned about exposure keratopathy, erosions, punctate keratopathy, epithelial defects and corneal infiltrate (Figure 2).29,30 In severe cases, this can lead to corneal thinning and subsequent perforation.29,30
Toxoplasmosis. While this can be contracted by anyone, it is particularly risky for pregnant patients because it can be transferred to the fetus during pregnancy. The parasitic disease caused by Toxoplasma gondii can be contracted via infected cat feces and eating undercooked contaminated meat, as well as from mother to child during pregnancy.19,31 Primary infection leads to congenital infection when it occurs during the pregnancy.
The timing of the infection during pregnancy will result in different levels of sequelae. First trimester fetal infection causes more severe complications, but most commonly the transmission occurs in the third trimester when the maternal and fetal circulation is greatest.19,31 Latent infections may become active during pregnancy, resulting in retinochoroiditis findings, and should be monitored and treated appropriately.19,31
Before either systemic or ocular treatment is initiated, clinicians should consult with the patient’s obstetrician. Systemic use of spiramycin is recommended over pyrimethamine as safer but equally effective.4
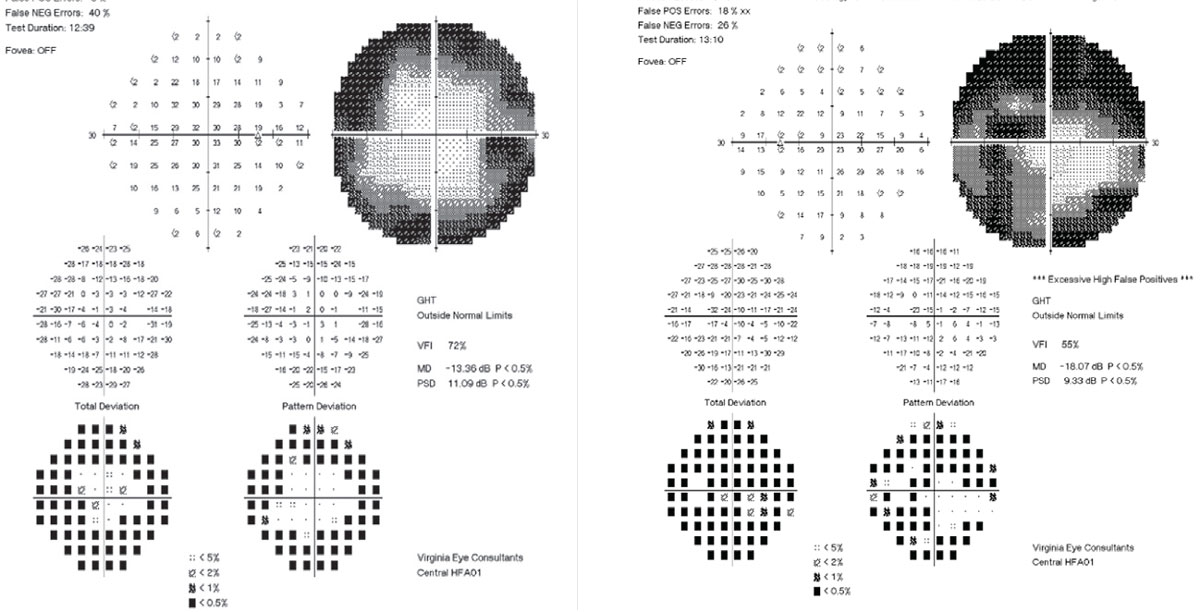 |
| Fig. 3. This previously diagnosed IIH patient was stable with no ONH swelling prior to pregnancy. During her pregnancy, her visual field, at left, shows constriction 360° with enlarged blind spot 360°, and her dilated exam revealed moderate ONH edema 360° OU. Because she was in her third trimester, consults with neurology and her obstetrician led to a plan to wait on any treatment and monitor closely for resolution after birth. Click image to enlarge. |
Central serous retinopathy. Neuro-sensory retinal detachment with associated retinal pigment epithelium (RPE) detachment, RPE leakage and RPE and choroidal hyper permeability is known as central serous retinopathy (CSR).19,31-33 Similar to non-pregnant patients, they will have complaints of decreased vision with unilateral or bilateral central metamorphopsia.
More often, pregnant women (90%) will have a subretinal exudate that is fibrous in nature compared with only 20% in their non-pregnant counterparts. This may be why, although the CSR resolves after pregnancy, it is more likely to reoccur in these patients.19,31-33 Treatment is not necessary in these patients, but they should be monitored closely.
Multiple sclerosis. Patients previously diagnosed with multiple sclerosis (MS) may note a decrease in attacks, including optic neuritis (ON), during pregnancy.19,31 However, there is an increase in occurrence of ON within the first three months postpartum.19,31 It is important that patients are aware of this so that they can report symptoms to their neurologist or optometrist quickly, ensuring a timely diagnosis and initiation of treatment, if indicated.
For these patients, pregnancy and delivery does not influence the patient’s mid or long-term disability as related to MS.31 It also doesn’t seem to influence the pregnancy, delivery or child’s health.31
Treatment is not indicated in every case of ON. For those whose visual acuity and field are not affected, the risk vs. benefit may not be worth initiating treatment. This requires consultation between the obstetrician, neurologist and optometrist. Treatment, for those who need it, both during pregnancy and while breastfeeding, is IV methylprednisolone for three days with an oral steroid taper of 1mg/kg/day for 11 days.34,35
Some physicians and optometrists do not follow the IV steroids with a oral steroid taper since there is no evidence suggesting efficacy of treatment is reliant on the oral taper.35 Other physicians use it as a way to control withdrawal side effects.35 Studies show that, although overall long-term visual outcome is the same as without treatment, it does delay the onset of clinically definite MS.34
Timing of treatment is also important; studies show that if steroids are started earlier in the course of ON it results in better outcomes and may help prevent vision loss compared with later treatment.36
Idiopathic intracranial hypertension (IIH). This overproduction of cerebral spinal fluid (CSF) is typically triggered by hormonal changes or weight gain, both of which occur during pregnancy (Figure 3).19,31,37 Ocular signs and symptoms include visual field defects, optic nerve head (ONH) elevation, diplopia, photopsia, headaches and tinnitus.19,31,37
For patients presenting with an edematous ONH without a diagnosis of IIH, testing is necessary to rule out alternative causes. An MRI and MRV with and without contrast of the head and orbits should be performed, followed by a lumbar puncture to confirm elevated CSF.
The standard treatment for IIH is oral medication Topamax (topiramate, Janssen Pharmaceuticals) or acetazolamide, which are historic category C. They may be considered if the obstetrician and neurologist determine that the benefits outweigh the risks.37 In most cases, patients are monitored closely with repeat visual fields and optical coherence tomography of the ONH every three months along with neurology or neuro-ophthalmology appointments.
If the patient becomes symptomatic with severe visual field defects, they may be treated with serial lumbar punctures throughout pregnancy, although that carries a risk of spontaneous abortion.19,37 In severe cases where vision is at risk, tube shunts or ONH fenestration may be needed.37
Glaucoma. Most of these medications are historic category C. Studies note that IOP decreases approximately 19.6% in pregnant patients with normal IOP and 24.4% in those with ocular hypertension.38-40 This occurs because of increased aqueous outflow, lower episcleral venous pressure due to decreased systemic vascular resistance and lower scleral rigidity resulting from increased tissue elasticity.38-40 The pressure returns to normal approximately two months postpartum.38-40
To effectively treat pregnant patients, clinicians must have a comprehensive understanding of how pregnancy can affect the ocular structures as well as the therapeutic options and associated risks. It is the optometrist’s job to help address patient concerns while providing safe, effective care.
Dr. Koetting is the referral optometric care and externship program coordinator at Virginia Eye Consultants in Norfolk, VA. She is a fellow of the AOA and a trustee of the Virginia Optometric Association.
1. Yenerel NM, Küçümen RB. Pregnancy and the Eye. Turk J Ophthalmol. 2015;45(5): 213-19. 2. U.S. Department of Health and Human Services. CHEMM FDA Pregnancy Categories. https://chemm.nlm.nih.gov/pregnancycategories.htm. 3. U.S. Food and Drug Administration. Pregnancy and Lactation Labeling (Drugs) Final Rule. www.fda.gov/drugs/labeling-information-drug-products/pregnancy-and-lactation-labeling-drugs-final-rule. 4. Bhatia J, Sadiq MN, Chaudhary TA, et al. Eye changes and risk of ocular medications during pregnancy and their management. Pak J Ophthalmol. 2007;23(1). 5. Chung CY, Kwok AK, Chung KL. Use of ophthalmic medications during pregnancy. Hong Kong Med J. 2004;10(3):191-5. 6. NIH Drugs and Lactation Database. www.ncbi.nlm.nih.gov/books/NBK501922. 7. FDA Center for Drug Evaluation and Research. www.accessdata.fda.gov/drugsatfda_docs/nda/2010/022272s000OtherR.pdf. 8. Xiidra Highlights of Prescribing Information. www.accessdata.fda.gov/drugsatfda_docs/label/2016/208073s000lbl.pdf. 9. Rhopressa Highlights of Prescribing Information. www.accessdata.fda.gov/drugsatfda_docs/label/2017/208254lbl.pdf. 10. Polizzi S, Mahajan VB. Intravitreal anti-VEGF injections in pregnancy: case series and review of literature. J Ocul Pharmacol Ther. 2015;31(10):605‐10. 11. Juncal VR, Paracha Q, Bamakrid M, et al. Ranibizumab and aflibercept levels in breast milk after intravitreal injection. Ophthalmol. 2020;127(2):278-80. 12. American Academy of Ophthalmology. Diagnostic Agents. www.aao.org/focalpointssnippetdetail.aspx?id=9c240b2e-6134-4926-a91d-6c69b1ee4a3d. 13. Rosales T, Isenberg S, Leake R, et al. Systemic effects of mydriatics in low weight infants. J Pediatr Ophthalmol Strabismus. 1981;18(6):42-4. 14. Fineman MS, Maguire JI, Fineman SW, et al. Safety of indocyanine green angiography during pregnancy: a survey of the retina, macula, and vitreous societies. Arch Ophthalmol. 2001;119(3):353-5. 15. Mattern J, Mayer PR. Excretion of fluorescein into breast milk. Am J Ophthalmol. 1990;109(5):598-9. 16. Mehdizadehkashi K, Chaichian S, Mehdizadehkashi A, et al. Visual acuity changes during pregnancy and postpartum: a cross-sectional study in Iran. J Pregnancy. 2014;2014:675792. 17. Sharma S, Rekha W, Sharma T, et al. Refractive issues in pregnancy. Aust N Z J Obstet Gynaecol. 2006;46(3):186-8. 18. Pizzarello LD. Refractive changes in pregnancy. Graefes Arch Clin Exp Ophthalmol. 2003;241(6):484-8. 19. Garg P, Aggarwal P. Ocular changes in pregnancy. Nepalese J Ophthalmol. 2012;4(1):150-61. 20. Schechter JE, Pidgeon M, Chang D, et al. Potential role of disrupted lacrimal acinar cells in dry eye during pregnancy. Adv Exp Med Biol. 2002;506(Pt A):153-7. 21. Sheth B, Mieler W. Ocular complications of pregnancy. Curr Opin Ophthalmol. 2001;12(6):455-63. 22. Vigil-De Gracia P, Ortega-Paz L. Retinal detachment in association with pre-eclampsia, eclampsia, and HELLP syndrome. Int J Gynecol Obstet. 2011;114(3):223-5. 23. Saito Y, Tano Y. Retinal pigment epithelial lesions associated with choroidal ischemia in pre-eclampsia. Retina. 1998;18(2):103-8. 24. Cunningham FG, Fernandez CO, Hernandez C. Blindness associated with preeclampsia and eclampsia. Am J Obstetr Gynecol. 1995;172(4):1291-8. 25. Centers for Disease Control and Prevention. Gestational Diabetes and Pregnancy. www.cdc.gov/pregnancy/diabetes-gestational.html. 26. American Optometric Association. Evidence Based Clinical Practice Guidelines: Eye care of the patient with Diabetes Mellitus. Second Edition. 2019. 27. Chang S, Fuhrmann M, Jovanovich L. The diabetes in early pregnancy study group (DIEP): Pregnancy, retinopathy normoglycemia. A preliminary analysis. Diabetes. 1985;35.Suppl:3A. 28. Vrabec, JT, Isaacson B, Hook JWV. Bell’s palsy and pregnancy. Otolaryngol Head Neck Surg. 2007;137(6):858-61. 29. Tiemstra JD, Khatkhate N. Bell’s palsy: diagnosis and management [Review]. Am Fam Physician. 2007;76(7):997-1002. 30. Lee V, Currie Z, Collin JR. Ophthalmic management of facial nerve palsy. Eye (Lond). 2004;18:1225-34. 31. Omoti AE, Waziri-Erameh JM, Okeigbemen VW. A review of the changes in the ophthalmic and visual system in pregnancy. Afr J Reprod Health. 2008;12(3):185-96. 32. Sunness JS, Haller JA, Fine SL. Central serous chorioretinopathy and pregnancy. Arch Ophthalmol. 1993;111(3):360-4. 33. Perkins SL, Kim JE, Pollack JS, et al. Clinical characteristics of central serous chorioretinopathy in women. Ophthalmology. 2002;109(2):262-6. 34. Optic Neuritis Study Group. Multiple sclerosis risk after optic neuritis: final optic neuritis treatment trial follow-up. Arch Neurol. 2008;65(6):727‐32. 35. Pula JH, Macdonald CJ. Current options for the treatment of optic neuritis. Clinical Ophthalmol. 2012;6:1211-23. 36. Plant GT, Sibtain NA, Thomas D. Hyperacute corticosteroid treatment of optic neuritis at the onset of pain may prevent visual loss: a case series. Mult Scler Int. 2011;2011:815068. 37. Huna-Baron R, Kupersmith MJ. Idiopathic intracranial hypertension in pregnancy. J Neurol. 2002;249(8):1078-81. 38. Horven I, Gjonnaess H. Corneal indentation pulse and intraocular pressure in pregnancy. Arch Ophthalmol. 1974;91(2):92-8. 39. Cantor LB, Harris A, Harris M. Glaucoma medications in pregnancy. Rev Ophthalmol. 2000;2000:91-9. 40. Johnson SM, Martinez M, Freedman S. Management of glaucoma in pregnancy and lactation. Surv Ophthalmol. 2001;45(5):449-54. 41. Alonso-Santander N, Ortega-Usobiaga J, Beltrán-Sanz J, et al. Laser in situ keratomileusis and surface ablation in breastfeeding patients. Cornea. January 30, 2020. [Epub ahead of print]. |
